- Search
| Ann Coloproctol > Volume 39(3); 2023 > Article |
|
See commentary "Organ preservation for early rectal cancer using preoperative chemoradiotherapy" in Volume 39 on page 191.
Abstract
Purpose
This study was designed to determine the feasibility of preoperative chemoradiotherapy (PCRT) in patients with clinical T2N0 distal rectal cancer.
Methods
Patients who underwent surgery for clinical T2N0 distal rectal cancer between January 2008 and December 2016 were included. Patients were divided into PCRT and non-PCRT groups. Non-PCRT patients underwent radical resection or local excision (LE) according to the surgeon’s decision, and PCRT patients underwent surgery according to the response to PCRT. Patients received 50.0 to 50.4 gray of preoperative radiotherapy with concurrent chemotherapy.
Results
Of 127 patients enrolled, 46 underwent PCRT and 81 did not. The mean distance of lesions from the anal verge was lower in the PCRT group (P=0.004). The most frequent operation was transanal excision and ultralow anterior resection in the PCRT and non-PCRT groups, respectively. Of the 46 patients who underwent PCRT, 21 (45.7%) achieved pathologic complete response, including 15 of the 24 (62.5%) who underwent LE. Rectal sparing rate was significantly higher in the PCRT group (11.1% vs. 52.2%, P<0.001). There were no significant differences in 3- and 5-year overall survival and recurrence-free survival regardless of PCRT or surgical procedures.
Graphical Abstract
Total mesorectal excision (TME) has become the reference standard of treatment for localized rectal cancer. Despite its efficacy in the control of local tumors, TME is associated with several complications and morbidities, including urinary and sexual dysfunction, defecatory problems, and permanent stoma [1, 2]. In addition, patients with far distal rectal cancer may require abdominoperineal resection (APR) when TME is performed. A watch and wait approach after preoperative chemoradiotherapy (PCRT) is an alternative to TME in selected patients, including those clinically suspected of achieving complete remission (CR) of the primary lesion without lymph node metastasis [3–6]. However, the future occurrence of CR cannot be predicted accurately, and high local recurrence rates have been reported [7–9]. Alternatives to TME include local excision (LE), such as transanal excision (TAE) or transanal microinvasive surgery (TAMIS); these may achieve pathologic CR (pCR) and can prevent complications such as low anterior resection (LAR) syndrome, as well as improve quality of life (QOL) without a permanent stoma [10].
LE may be indicated in patients with early, clinical T1N0 rectal cancer without high-risk features such as lymphovascular invasion or perineural invasion [11]. The performance of LE precludes the performance of lymph node dissection, and LE alone is associated with a higher rate of local recurrence than TME [12].
PCRT followed by TME has become the standard of treatment in patients with clinical T3–4 and/or node-positive patients, and it has shown good local control in several randomized controlled trials (RCTs) [13–15]. As an extension of these findings, several studies found that PCRT before LE may be effective for local tumor control, and PCRT followed by LE may have oncologic outcomes similar to those of TME [16, 17]. Approximately 8% to 24% of patients show pCR after PCRT [18], whereas the oncologic outcomes of LE in patients with a good response to PCRT are similar to those of TME [16, 19, 20].
The standard treatment of patients with clinical T2N0 rectal cancer is radical surgery with TME. The clinical benefits of PCRT in these patients are not widely recognized. In this study, we retrospectively assessed the feasibility of PCRT in patients with clinical T2N0 rectal cancer and analyzed the outcomes of PCRT, including its likelihood of rectal preservation.
This study protocol was approved by the Institutional Review Board of Asan Medical Center (No. 2020-0967). The requirement for informed consent was waived because of the retrospective nature of the study.
The medical records of patients with clinical T2N0 distal rectal cancer who underwent surgery between January 2008 and December 2016 were retrospectively reviewed. Patients were staged according to endorectal ultrasound, magnetic resonance imaging, and colonoscopy results. Patients were included if they had pathologically confirmed adenocarcinoma within 4 cm of the anal verge (AV) and did not have distant metastases.
Patients included in the study were divided into 2 groups, those who underwent PCRT and those who did not. Upfront surgery was mainly performed in patients who were less likely to undergo APR considering sex, length of the anal canal, and pelvic diameter. Among these patients, those clinically likely to have < T2 disease underwent LE based on the surgeon’s opinion. Patients who were considered more likely to undergo APR based on clinical factors underwent PCRT to reduce the tumor size and avoid APR.
Patients received total 50.0 to 50.4 Gy of preoperative radiotherapy, which was administered as 45.0 to 46.0 Gy to the entire pelvis in 1.8 to 2.0 Gy daily fractions, followed by a 4.0 to 5.4 Gy boost to the primary tumor in a 1.8 to 2.0 Gy daily fractions. Patients received concurrent chemotherapy consisting of oral capecitabine (825 mg/m2, twice daily) or 2 cycles of 5-fluorouracil (375 mg/m2/day, for 3 days) with leucovorin (20 mg/m2/day, for 3 days) during the 1st and 5th weeks of radiotherapy.
Patients who underwent PCRT had surgery 7 to 8 weeks after PCRT. Patients in the TME group underwent ultralow anterior resection (uLAR); an operation to remove part of the left side of the colon including the entire rectum; or APR, whereas patients in the LE group underwent TAE or TAMIS, a method of excising full-thickness tumors.
All patients were followed up for approximately 5 years after surgery, after which patients were recommended to undergo regular medical examinations every 2 years, including laboratory tests such as serum carcinoembryonic antigen measurement, abdominopelvic computed tomography (APCT), chest computed tomography (CCT), and colonoscopy. All patients underwent surveillance every 6 months at the outpatient clinic. Patients in both the TME and LE groups underwent APCT every 6 months and CCT every 6 to 12 months. Patients in both groups were evaluated by colonoscopy 6 months after surgery; patients in the TME group underwent colonoscopy every 2 to 3 years thereafter, whereas patients in the LE group underwent alternate sigmoidoscopy and colonoscopy every 6 months.
Categorical variables were compared by the chi-square test and continuous variables by the Student t-test. Overall survival (OS) and disease-free survival (DFS) were analyzed by the Kaplan-Meier method and compared by the log-rank test. All statistical analyses were performed using IBM SPSS ver. 25.0 (IBM Corp), and a P-value of < 0.05 indicated statistical significance.
This study enrolled 127 patients diagnosed with clinical T2N0 rectal cancer. Among them, 46 patients underwent PCRT and 81 patients did not (Fig. 1). The demographic and clinical characteristics of the patients at baseline and the clinicopathological features of the tumors are shown in Table 1. The mean distance from the AV to the tumor was approximately 3 cm in both groups, although that of patients in the PCRT group was shorter (2.88±1.09 cm vs. 3.36±0.75 cm, P=0.004). TAE was the most common surgery in the PCRT group (43.5%), whereas uLAR was the most common operation in patients who did not undergo PCRT (81.5%). Rectal sparing rate via LE (TAE, TAMIS) was significantly higher in the PCRT group (11.1% vs. 52.2%, P<0.001). The proportion of patients who underwent APR was significantly higher in the PCRT group (21.7% vs. 7.4%, P<0.001). The proportion of men was higher in the PCRT group, although the difference was not statistically significant (71.7% vs. 55.6%, P=0.089). And there was no difference in mean age between the 2 groups (61.87±11.01 years vs. 61.20±12.91 years, P=0.757). More than half of the patients in both groups had moderately differentiated tumors, and R0 resection was performed in all patients. Approximately 45% of patients who underwent PCRT achieved pCR (ypT0); in the upfront surgery group, 15 patients were finally diagnosed as pathologic T1 category and 2 patients were T3 category.
Table 2 shows the characteristics of the patients according to the surgical methods. Among the 46 patients who underwent PCRT, 24 underwent LE and 22 underwent TME. Fifteen of the 24 patients (62.5%) who underwent LE after PCRT achieved ypT0. Of the 22 patients in the TME after PCRT group, 6 (27.3%) achieved ypT0, and 8 (36.4%) each had ypT1 and ypT2. The percentage of patients with ypT0 was significantly higher in the LE than in the TME group after PCRT (P=0.042). Three patients in the PCRT group were confirmed to have ypT2 stage disease after LE. Although radical surgery was recommended for oncologic safety, none of these patients underwent radical surgery because of old age, poor general performance, and patient’s refusal in each of the 3 patients. These patients all received adjuvant chemotherapy rather than radical surgery. One patient experienced local and systemic recurrence and died of cancer-related causes. There were no significant differences in median age (64.0 years vs. 61.5 years, P=0.191), tumor distance from the AV (2.73±1.24 cm vs. 3.05±0.90 cm, P=0.275), and the differentiation of tumors (P=0.958) between the 2 groups. Among the 81 patients in non-PCRT group, 9 underwent LE and 72 underwent TME. The rate of adjuvant treatment such as chemotherapy and radiotherapy was significantly higher in the LE without PCRT group (P<0.001). Two patients in the non-PCRT group were confirmed as pT2 stage after LE. One patient was recommended for radical surgery, but the patient refused. The patient had adjuvant concurrent chemoradiotherapy and survived without recurrence during the 5-year follow-up. The other patient underwent neoadjuvant chemoradiotherapy before radical surgery but refused radical surgery after neoadjuvant chemoradiotherapy. The patient experienced local recurrence after 29 months of LE and underwent salvage surgery. He is alive without recurrence after salvage surgery. There were no significant differences in age, sex, median distance from the AV, and pathologic characteristics between the LE and TME in the nonPCRT group.
The median follow-up was 59.0 months (range, 50.5–69.0 months) and there were no differences between the LE and TME groups (P=0.225); the follow-up period was 59.5 months (range, 39.0–70.3 months) in the LE (TAE, TAMIS) group and 59.0 months (range, 53.0–69.0 months) in the TME (uLAR, APR) group.
Fig. 2 shows recurrence-free survival (RFS) rates of included patients according to PCRT and surgical methods. The mean RFS did not differ according to PCRT (PCRT, 131.71±5.47 months vs. nonPCRT, 133.85±4.38 months; P=0.775) or surgical methods (TME, 133.69±4.18 months vs. LE, 113.74±7.04 months; P=0.327). The 3-year RFS rates in patients who underwent PCRT were 95.8% in the LE after PCRT group and 90.9% in the TME after PCRT group (P=0.50). Supplementary Table 1 shows information about patients with recurrent tumors. Among the patients who underwent PCRT, 1 patient in the LE group and 2 patients in the TME group developed distant metastasis. All 3 patients who experienced systemic recurrence before 3 years in the PCRT group underwent salvage surgery and all remain alive without any evidence of disease. The 3-year RFS rates in the non-PCRT group were not significantly different according to the surgical method (LE, 77.8% vs. TME, 93.1%; P=0.183). Among 6 patients who developed local recurrence in the non-PCRT group, 5 patients had salvage operations and were alive without evidence of recurrence at the time of this publication. One patient who underwent stereotactic body radiation therapy and chemotherapy after pelvic sidewall recurrence was alive with stable disease. And 1 patient underwent palliative chemotherapy after systemic metastasis including liver, lung, and bone expired at 59 months after primary surgery.
The 5-year survival rate was analyzed in patients who were followed up for more than 5 years, 39 in the PCRT group and 32 in the non-PCRT group. In patients who underwent PCRT, the 5-year RFS rates were not significantly different between LE and TME (LE, 83.3% vs. TME, 89.5%; P=0.59). Two additional patients in the PCRT group experienced recurrences 3 years after surgery and all of them underwent LE as primary surgery. One patient presented with multiple lung metastases and local pelvic sidewall recurrence. He was treated with 8 cycles of chemotherapy with FOLFOX (folinic acid, fluorouracil, and oxaliplatin) plus bevacizumab, but he showed disease progression. He was subsequently treated with 8 cycles of additional chemotherapy with FOLFIRI (folinic acid, fluorouracil, and irinotecan) plus bevacizumab, but continued to show disease progression. Finally, he died 5 years and 6 months after surgery. The other patient showed local pelvic sidewall recurrence 39 months after surgery. He initially underwent TAMIS for rectal cancer 4 cm from the AV. The tumor was ypT2 category with clear resection margins, but the patient refused radical surgery. He underwent 4 cycles of adjuvant chemotherapy with the leucovorin and fluorouracil regimen. Three years later, a 5 cm left pelvic sidewall mass was detected and R1 resection was done. And the patient was treated with 24 cycles of chemotherapy with FOLFIRI and bevacizumab, followed by 4 cycles of chemotherapy with FOLFOX and bevacizumab. However, his disease continued to progress with the development of lung metastasis. The patient died 5 years and 3 months after the first operation. The 5-year RFS rates of the non-PCRT group were not significantly different between LE and TME (LE, 66.6% vs. TME, 93.1%; P=0.263). No additional patient experienced recurrence after 3 years in the non-PCRT group. The 5-year OS rates were 100% in the PCRT group and 98.9% in the non-PCRT group.
Complications caused by PCRT were defined as symptoms that occurred in the period between the start of PCRT and before surgery. PCRT was associated with a complication rate of 45.8% in the LE after PCRT group and 45.5% in the TME after PCRT group (P=0.979). All complications belonged to Clavien-Dindo classification I, and it was treated only by symptom control through the administration of analgesics or antidiarrheal drugs as needed. Anal pain was the most frequent complication in both groups (37.5% vs. 31.8%) (Supplementary Table 2).
Most of complications except one who experienced hand-foot syndrome (HFS), improved within 1 month after the end of PCRT. The duration of symptoms of HFS experienced by 1 patient was not recorded, but no additional medications were administered after PCRT.
Acute surgical complications were defined as symptoms that occurred within 1 month after surgery. There was no significant relationship between the surgical complication rate and PCRT (P=0.410). The incidence of complications such as wound problems and mechanical ileus may have been higher in the TME group, although the statistical significance was difficult to determine because of the small number of events.
Of 18 patients who experienced surgical complications, 6 were in the PCRT group and 12 were in the upfront surgery group. Among patients who underwent PCRT, 1 patient in the LE after PCRT group underwent examination under anesthesia because of a perianal abscess. The previous TAE closure site had been disrupted, and incision and drainage were performed. Five patients in the TME after PCRT group experienced surgical complications. Two experienced prolonged ileus, whereas the other 3 had wound problems requiring further management. Two of these 3 patients were treated with antibiotics, and 1 underwent bedside wound repair. Among patients who underwent upfront surgery, there were no surgical complications in the LE without PCRT group, whereas 12 patients in the TME without PCRT group experienced surgical complications. Seven patients experienced mechanical ileus, wound problems, voiding difficulty, and postoperative syndrome of inappropriate antidiuretic hormone, and were treated with supportive care; the other 5 patients suffered anastomosis leakage, pelvic hematoma, and parastomal hernia, and were treated with reoperations. None of the patients who experienced complications died (Supplementary Table 3).
PCRT followed by TME improves local control in patients with locally advanced rectal cancer and is regarded as a standard treatment in many countries [21, 22]. Improved oncologic outcomes have been reported in patients with advanced rectal cancer who achieved a good response to PCRT [23, 24]. In a study, the 5-year RFS of patients who achieved pCR after PCRT was comparable to that of patients with tumors confined to the rectal mucosa [23].
Because of the absence of a residual tumor in ypT0 patients, surgery may have no advantages over observation alone, except for confirming pathologic stage [4]. Moreover, surgery for distal rectal cancer is associated with mortality and morbidities, including anastomosis leak, sexual and urinary dysfunction, wound infection, and diverting or permanent ostomy formation [25–27]. Based on these results, observation alone was tested in highly selected patients who achieved clinical complete response (cCR) to PCRT [4]. Surgery and observation showed similar long-term outcomes in patients with stage 0 disease, with cCR rates increasing from 27% to 49.2% between 1998 and 2014 [3–6]. The local recurrence rate was > 30%, and approximately 93% of patients with recurrence were eligible for salvage surgery [6]. The 5-year cancer-specific OS and DFS rates were 91% and 68%, respectively [6]. Other studies, however, did not show similarly good oncologic outcomes, with cCR rates ranging from 0% to 25%, and local recurrence rates from 5% to 50% [7–9, 28]. These discrepancies raise concerns about the oncological safety of observation alone [7, 29]. Furthermore, there is no reliable preoperative imaging modality that can predict pCR [24, 30]. Thus, LE with full thickness is increasingly performed to assure CR and organ preservation [10].
The main drawback to LE after PCRT as an organ-preserving treatment is that lymph node dissection cannot be performed. Many studies failed to find differences in the oncological outcomes of patients who underwent TME and LE when ypT0-1 was expected because of major tumor regression [16, 19, 20]. Most of these studies reported OS rates of 85% to 100% and local recurrence rates of 3% to 9% after LE [16, 19, 20, 31]. A multi-institutional phase 2 trial of patients with clinical T2N0 distal rectal cancer (ACOSOG Z6041) who underwent PCRT followed by LE found that the local and systemic recurrence rates were 5.1% and 6.3%, respectively [24]. These results were superior to those of a population-based analysis from the National Cancer Database and the Surveillance, Epidemiology and End Results database, which reported a 5-year OS rate close to 76% in patients with clinical T2N0 rectal cancer who underwent transabdominal resection alone [32]. These findings suggest that radical surgery can be avoided in selected patients who respond well to PCRT [24].
Studies have reported that there are no differences in surgical complications between patients who undergo LE and those who undergo TME [33–35]. However, functional QOL, as determined by the rates of rectal sparing and avoiding stoma, are better in those who undergo LE [36, 37]. Currently, an RCT comparing LE and TME in patients with mid-to-low rectal cancer who have magnetic resonance tumor regression grade 1 or 2 after PCRT is ongoing [38].
Although various surgical techniques and PCRT have enabled sphincter-saving resection in patients with distal rectal cancer, APR may provide better oncologic outcomes in some patients. However, APR results in a permanent stoma and poorer QOL. In principle, TME should be performed in patients with cT2N0 rectal cancer, whereas APR should be infrequent. This study, therefore, attempted to determine the percentage of cT2N0 patients who can avoid APR and undergo sphincter saving such as uLAR or rectal saving surgery which preserve rectal volume such as TAE or TAMIS if they have a good response to PCRT. Approximately 87.4% of the patients in this study had sphincter-saving surgery, which enabled 78.3% in the PCRT group and 92.6% in the non-PCRT group to avoid APR. Moreover, the rectum of 52.2% of patients in the PCRT group and 11.1% in the non-PCRT group could be preserved via LE, and 45.7% of patients were pathologically confirmed as ypT0 after surgery in the PCRT group. The rectal saving rate was significantly higher in the PCRT group (P<0.001), whereas the sphincter-saving rate was higher in the non-PCRT group (P=0.022). This may be attributed to the fact that PCRT was performed to avoid APR in patients with high APR potential when underwent TME due to tumor location, size, sex, length of the anal canal, and anatomical problems such as narrow pelvis.
The mean distance from the AV was significantly longer in the upfront surgery group than in the PCRT group. This indicates that upfront surgery was attempted in patients who had a high probability of sphincter saving despite TME. Patient and tumor characteristics were similar between the LE and TME groups regardless of PCRT. In the PCRT group, the number of patients who achieved pCR was higher in the LE group. In addition, 33% of patients who underwent LE were confirmed to be pathologic T1 in the upfront surgery group. This may be because LE was preferentially performed according to the surgeon’s decision in patients who had focal T2 suspicion, but in whom the possibility of T1 could not be excluded.
There were no statistically significant differences in 3- and 5-year OS and RFS between the PCRT group and the upfront surgery group. Although not statistically significant, RFS was lower in patients who underwent LE as upfront surgery; this was thought to be due to the small number of patients who underwent LE as upfront surgery and the fact that most of them refused radical surgery to lower the recurrence rate. In patients who underwent PCRT, the difference in mean cancer-specific survival between the LE and TME groups was marginally significant (84.6±5.4 months vs. 100.5±5.8 months, P=0.05), which is likely because the mean follow-up period was approximately 10 months longer in the TME than in the LE group.
Patients who undergo PCRT may experience treatment-related complications that do not occur in patients who undergo surgery immediately after diagnosis. Although approximately 50% of patients who underwent PCRT experienced complications, there were no complications requiring additional treatment compared to the non-PCRT group.
This study had several limitations. First, the retrospective design of the study resulted in selection bias; decisions regarding the surgical procedure and the administration of PCRT were not determined in a randomized manner, and were instead based on the operator’s subjective judgment according to the results of examinations. Because LE was performed in patients with a good response to PCRT or in those thought to be lower than T2 according to the operator’s judgment, the pathologic stage was lower in the LE group, and the proportion of patients who achieved pCR was significantly higher in the LE than in the TME group. However, the overall pCR rate was comparable with that reported in previous prospective trials [24, 39] including ACOSOG Z6041. Second, after undergoing upfront surgery, 33.3% of clinical T2N0 patients showed pathological T1 stage. This result suggests that it was difficult to accurately evaluate clinical T2N0 only by preoperative imaging studies, and it is possible that some of the pathologic T1N0 patients were included even among patients who underwent PCRT. Therefore, the present patient population may be representative of clinical T2N0 patients, unlike other studies that only enrolled patients who underwent LE after PCRT. Third, the number of patients enrolled in this study was small because the standard treatment of cT2N0 rectal cancer is upfront surgery with TME rather than PCRT or LE. Fourth, QOL and functional outcomes after surgery were not analyzed in this study. Prospective trials comparing cT2N0 patients according to PCRT status and surgical procedures and the inclusion of a larger number of patients are needed. In addition, future studies should compare functional outcomes, including urinary dysfunction, incontinence, and sexual dysfunction, after surgery.
The results of this study suggest that PCRT can increase the rate of rectal sparing via LE in selected patients with clinical T2N0 distal rectal cancer. LE alone can lead to good oncologic outcomes in approximately 45% of patients with ypT0.
In conclusion, PCRT in clinical T2N0 distal rectal cancer patients is an effective alternative treatment that can improve rectal sparing rates via LE with acceptable oncological outcomes when clinically highly suspected to do APR.
SUPPLEMENTARY MATERIALS
Supplementary materials for this study are presented online (available at https://doi.org/10.3393/ac.2022.00066.0009).
Supplementary Table 1.
Characteristics of patients who experienced local and distant recurrences
Supplementary Table 2.
Complications of preoperative chemoradiotherapy
Supplementary Table 3.
Surgical complications in the study patients according to type of surgery
Fig. 1.
Flowchart of included patients. AV, anal verge; PCRT, preoperative chemoradiotherapy; TME, total mesorectal excision.
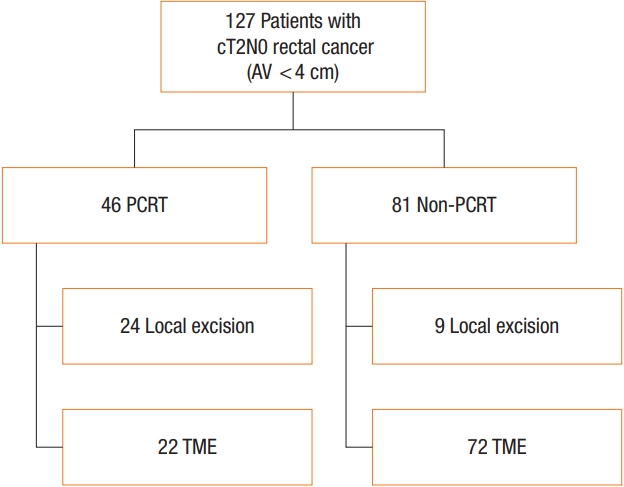
Fig. 2.
Kaplan-Meier estimates of recurrence-free survival of patients. (A) Recurrence-free survival (RFS) in the preoperative chemoradiotherapy (PCRT) and non-PCRT groups. (B) RFS in the total mesorectal excision (TME) and local excision (LE) groups. (C) RFS according to
PCRT and surgical procedures.
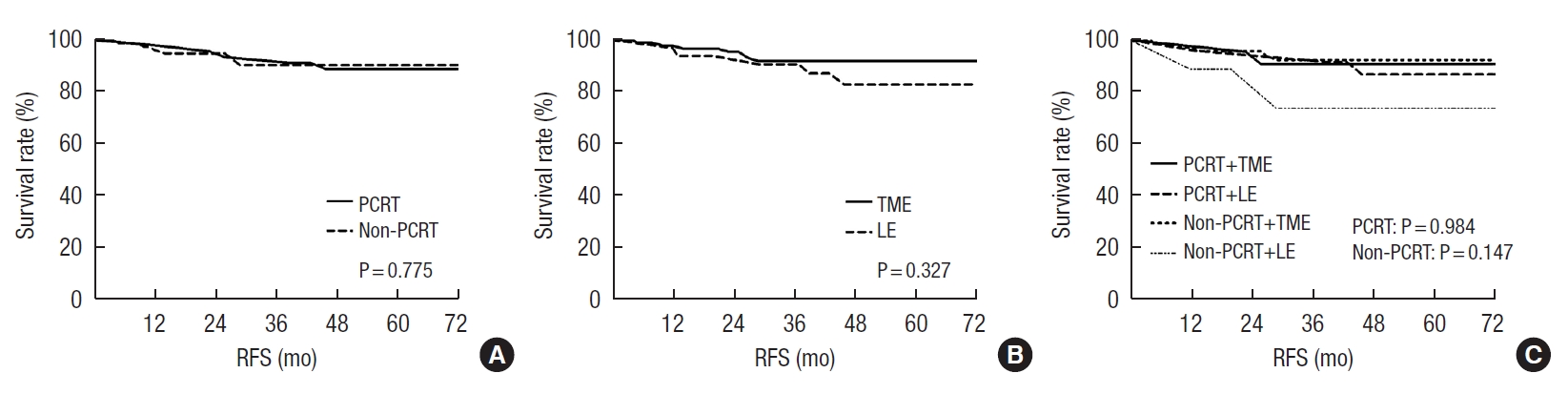
Table 1.
Demographic and clinicopathological characteristics of the study patients (n=127)
Values are presented as mean±standard deviation or number (%).
PCRT, preoperative chemoradiotherapy; APR, abdominoperineal resection; uLAR, ultralow anterior resection; TAE, transanal excision; TAMIS, transanal minimally invasive surgery; WD, well-differentiated; MD, moderately differentiated; PD, poorly differentiated; NA, not applicable.
Table 2.
Demographic and clinicopathological characteristics of patients according to type of surgery
REFERENCES
1. Chessin DB, Enker W, Cohen AM, Paty PB, Weiser MR, Saltz L, et al. Complications after preoperative combined modality therapy and radical resection of locally advanced rectal cancer: a 14-year experience from a specialty service. J Am Coll Surg 2005;200:876–84.


2. Peeters KC, van de Velde CJ, Leer JW, Martijn H, Junggeburt JM, Kranenbarg EK, et al. Late side effects of short-course preoperative radiotherapy combined with total mesorectal excision for rectal cancer: increased bowel dysfunction in irradiated patients: a Dutch colorectal cancer group study. J Clin Oncol 2005;23:6199–206.


3. Habr-Gama A, de Souza PM, Ribeiro U Jr, Nadalin W, Gansl R, Sousa AH Jr, et al. Low rectal cancer: impact of radiation and chemotherapy on surgical treatment. Dis Colon Rectum 1998;41:1087–96.

4. Habr-Gama A, Perez RO, Nadalin W, Sabbaga J, Ribeiro U Jr, Silva e Sousa AH Jr, et al. Operative versus nonoperative treatment for stage 0 distal rectal cancer following chemoradiation therapy: long-term results. Ann Surg 2004;240:711–8.


5. Habr-Gama A, Perez RO, Proscurshim I, Campos FG, Nadalin W, Kiss D, et al. Patterns of failure and survival for nonoperative treatment of stage c0 distal rectal cancer following neoadjuvant chemoradiation therapy. J Gastrointest Surg 2006;10:1319–29.


6. Habr-Gama A, Gama-Rodrigues J, São Julião GP, Proscurshim I, Sabbagh C, Lynn PB, et al. Local recurrence after complete clinical response and watch and wait in rectal cancer after neoadjuvant chemoradiation: impact of salvage therapy on local disease control. Int J Radiat Oncol Biol Phys 2014;88:822–8.


7. Glynne-Jones R, Hughes R. Critical appraisal of the ‘wait and see’ approach in rectal cancer for clinical complete responders after chemoradiation. Br J Surg 2012;99:897–909.


8. Dalton RS, Velineni R, Osborne ME, Thomas R, Harries S, Gee AS, et al. A single-centre experience of chemoradiotherapy for rectal cancer: is there potential for nonoperative management? Colorectal Dis 2012;14:567–71.


9. Smith JJ, Strombom P, Chow OS, Roxburgh CS, Lynn P, Eaton A, et al. Assessment of a watch-and-wait strategy for rectal cancer in patients with a complete response after neoadjuvant therapy. JAMA Oncol 2019;5:e185896.



10. Creavin B, Ryan E, Martin ST, Hanly A, O’Connell PR, Sheahan K, et al. Organ preservation with local excision or active surveillance following chemoradiotherapy for rectal cancer. Br J Cancer 2017;116:169–74.



11. Doornebosch PG, Zeestraten E, de Graaf EJ, Hermsen P, Dawson I, Tollenaar RA, et al. Transanal endoscopic microsurgery for T1 rectal cancer: size matters! Surg Endosc 2012;26:551–7.


12. Nascimbeni R, Burgart LJ, Nivatvongs S, Larson DR. Risk of lymph node metastasis in T1 carcinoma of the colon and rectum. Dis Colon Rectum 2002;45:200–6.


13. Sauer R, Becker H, Hohenberger W, Rödel C, Wittekind C, Fietkau R, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 2004;351:1731–40.


14. Cammà C, Giunta M, Fiorica F, Pagliaro L, Craxì A, Cottone M. Preoperative radiotherapy for resectable rectal cancer: a metaanalysis. JAMA 2000;284:1008–15.


15. Swedish Rectal Cancer Trial. Cedermark B, Dahlberg M, Glimelius B, Påhlman L, Rutqvist LE, et al. Improved survival with preoperative radiotherapy in resectable rectal cancer. N Engl J Med 1997;336:980–7.


16. Pucciarelli S, De Paoli A, Guerrieri M, La Torre G, Maretto I, De Marchi F, et al. Local excision after preoperative chemoradiotherapy for rectal cancer: results of a multicenter phase II clinical trial. Dis Colon Rectum 2013;56:1349–56.

17. Jung SM, Yu CS, Park IJ, Kim TW, Kim JH, Yoon YS, et al. Oncologic safety of local excision compared with total mesorectal excision for ypT0-T1 rectal cancer: a propensity score analysis. Medicine (Baltimore) 2016;95:e3718.



18. Maas M, Nelemans PJ, Valentini V, Das P, Rödel C, Kuo LJ, et al. Long-term outcome in patients with a pathological complete response after chemoradiation for rectal cancer: a pooled analysis of individual patient data. Lancet Oncol 2010;11:835–44.


19. Yu CS, Yun HR, Shin EJ, Lee KY, Kim NK, Lim SB, et al. Local excision after neoadjuvant chemoradiation therapy in advanced rectal cancer: a national multicenter analysis. Am J Surg 2013;206:482–7.


20. Kundel Y, Brenner R, Purim O, Peled N, Idelevich E, Fenig E, et al. Is local excision after complete pathological response to neoadjuvant chemoradiation for rectal cancer an acceptable treatment option? Dis Colon Rectum 2010;53:1624–31.


21. van de Velde CJ, Boelens PG, Borras JM, Coebergh JW, Cervantes A, Blomqvist L, et al. EURECCA colorectal: multidisciplinary management: European consensus conference colon & rectum. Eur J Cancer 2014;50:1.

22. Hashiguchi Y, Muro K, Saito Y, Ito Y, Ajioka Y, Hamaguchi T, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol 2020;25:1–42.



23. Hwang K, Park IJ, Yu CS, Lim SB, Lee JL, Yoon YS, et al. Impression of prognosis regarding pathologic stage after preoperative chemoradiotherapy in rectal cancer. World J Gastroenterol 2015;21:563–70.



24. Garcia-Aguilar J, Renfro LA, Chow OS, Shi Q, Carrero XW, Lynn PB, et al. Organ preservation for clinical T2N0 distal rectal cancer using neoadjuvant chemoradiotherapy and local excision (ACOSOG Z6041): results of an open-label, single-arm, multi-institutional, phase 2 trial. Lancet Oncol 2015;16:1537–46.



25. Petrelli NJ, Nagel S, Rodriguez-Bigas M, Piedmonte M, Herrera L. Morbidity and mortality following abdominoperineal resection for rectal adenocarcinoma. Am Surg 1993;59:400–4.

26. Maas CP, Moriya Y, Steup WH, Kiebert GM, Kranenbarg WM, van de Velde CJ. Radical and nerve-preserving surgery for rectal cancer in The Netherlands: a prospective study on morbidity and functional outcome. Br J Surg 1998;85:92–7.


27. Nesbakken A, Nygaard K, Bull-Njaa T, Carlsen E, Eri LM. Bladder and sexual dysfunction after mesorectal excision for rectal cancer. Br J Surg 2000;87:206–10.


28. Ayloor Seshadri R, Kondaveeti SS, Jayanand SB, John A, Rajendranath R, Arumugam V, et al. Complete clinical response to neoadjuvant chemoradiation in rectal cancers: can surgery be avoided? Hepatogastroenterology 2013;60:410–4.

29. Minsky BD. Rectal cancer: is ‘watch and wait’ a safe option for rectal cancer? Nat Rev Gastroenterol Hepatol 2013;10:698–700.



30. Hiotis SP, Weber SM, Cohen AM, Minsky BD, Paty PB, Guillem JG, et al. Assessing the predictive value of clinical complete response to neoadjuvant therapy for rectal cancer: an analysis of 488 patients. J Am Coll Surg 2002;194:131–6.


31. Nair RM, Siegel EM, Chen DT, Fulp WJ, Yeatman TJ, Malafa MP, et al. Long-term results of transanal excision after neoadjuvant chemoradiation for T2 and T3 adenocarcinomas of the rectum. J Gastrointest Surg 2008;12:1797–806.


32. You YN, Baxter NN, Stewart A, Nelson H. Is the increasing rate of local excision for stage I rectal cancer in the United States justified?: a nationwide cohort study from the National Cancer Database. Ann Surg 2007;245:726–33.


33. Lezoche G, Baldarelli M, Guerrieri M, Paganini AM, De Sanctis A, Bartolacci S, et al. A prospective randomized study with a 5-year minimum follow-up evaluation of transanal endoscopic microsurgery versus laparoscopic total mesorectal excision after neoadjuvant therapy. Surg Endosc 2008;22:352–8.


34. Rullier E, Rouanet P, Tuech JJ, Valverde A, Lelong B, Rivoire M, et al. Organ preservation for rectal cancer (GRECCAR 2): a prospective, randomised, open-label, multicentre, phase 3 trial. Lancet 2017;390:469–79.


35. Lezoche E, Baldarelli M, Lezoche G, Paganini AM, Gesuita R, Guerrieri M. Randomized clinical trial of endoluminal locoregional resection versus laparoscopic total mesorectal excision for T2 rectal cancer after neoadjuvant therapy. Br J Surg 2012;99:1211–8.



36. Martens MH, Maas M, Heijnen LA, Lambregts DM, Leijtens JW, Stassen LP, et al. Long-term outcome of an organ preservation program after neoadjuvant treatment for rectal cancer. J Natl Cancer Inst 2016;108:djw171.


37. Doornebosch PG, Gosselink MP, Neijenhuis PA, Schouten WR, Tollenaar RA, de Graaf EJ. Impact of transanal endoscopic microsurgery on functional outcome and quality of life. Int J Colorectal Dis 2008;23:709–13.



- TOOLS