- Search
| Ann Coloproctol > Volume 38(5); 2022 > Article |
|
See commentary "Can the cross-sectional area of the psoas muscle be a predictor of anastomotic failure in male rectal cancer patients?" in Volume 38 on page 333.
Abstract
Purpose
Preoperative sarcopenia worsens postoperative outcomes in various cancer types including colorectal cancer. However, we often experienced postoperative anastomotic leakage in muscular male patients such as Judo players, especially in rectal cancer surgery with lower anastomosis. It is controversial whether the whole skeletal muscle mass impacts the potential for anastomotic failure in male rectal cancer patients. Thus, the purpose of this study was to clarify whether skeletal muscle mass impacts anastomotic leakage in rectal cancer in men.
Methods
We reviewed the medical charts of male patients suffering from rectal cancer who underwent colo-procto anastomosis below the peritoneal reflection without a protective diverting stoma. We measured the psoas muscle area and calculated the psoas muscle index.
Results
One hundred ninety-seven male rectal cancer patients were enrolled in this study. The psoas muscle index was significantly higher in patients with anastomotic leakage (P<0.001). Receiver operating characteristic curve determined the optimal cut-off value of the psoas muscle index for predicting anastomotic leakage as 812.67 cm2/m2 (sensitivity of 60% and specificity of 74.3%). Multivariate analysis revealed that high psoas muscle index (risk ratio [RR], 3.933; P<0.001; 95% confidence interval [CI], 1.917–8.070) and super low anastomosis (RR, 2.792; P=0.015; 95% CI, 1.221–6.384) were independent predictive factors of anastomotic leakage.
Graphical Abstract
Colorectal cancer (CRC), the third most common cancer worldwide, accounting for approximately 10% of total cancer incidence, predominantly occurs in developed countries [1]. In Japan, CRC is the leading cause of death in women and the third in men; over 130,000 patients were newly diagnosed with CRC in 2014, and over 50,000 patients died from CRC in 2017 [2]. Surgical resection is the mainstay curative treatment for CRC patients. Innovations in surgical procedures, such as laparoscopic surgery [3], are important in improving CRC patients’ treatment outcomes and prognoses.
In rectal cancer surgery, the most frequent and difficult postoperative adverse events are associated with the anastomotic site, such as anastomotic leakage (AL) and anastomotic site stenosis. Especially for AL, it is most important for colorectal surgeons to avoid this complication because AL can be postoperative mortality, and the leakage rate after low anterior resection has been reported to range from 4% to 29%. Furthermore, many studies have reported that AL also affects the long-term prognosis in rectal cancer [4-6]. Thus, avoiding AL in rectal cancer surgery could improve the long-term outcome of rectal cancer patients. To attempt to reduce this difficult complication, many surgeons perform a diverting stoma (DS) for high-risk patients with comorbidities such as diabetes mellitus, arteriosclerosis, and steroid usage [7]. A DS can worsen the body image and quality of life of the patients at least temporarily [8], and another surgery is required to close the stoma. Recent studies reported that the incidence of AL can be decreased by fluorescent blood flow evaluation using indocyanine green [9, 10], and a transanal decompression tube for anastomosis protection can reduce the pressure on the anastomotic site and led to decreased AL [11], but this is not yet well standardized.
A previous meta-analysis showed that male sex, high body mass index (BMI), lower anastomosis, longer surgery time, high volume of estimated blood loss, number of stapler firings over 3 times, and preoperative chemotherapy were found to be significantly associated with the development of AL [12, 13]. More recently, the concept of sarcopenia has gained attention in the field of clinical research, which includes surgical oncology. Sarcopenia is defined as the loss of skeletal muscle mass and strength with increasing risk of physical disability, immunodeficiency, poor quality of life, and mortality. Previous studies have shown that preoperative sarcopenia worsens both short- and long-term outcomes in various types of resectable cancers [14-18]. In rectal cancer, preoperative low skeletal muscle mass was reported to be a predictive marker of poor outcome [19]. Previous studies have shown that whole skeletal muscle mass, an indicator of sarcopenia, is correlated with psoas muscle index (PMI), which is corrected by height [20]. Another study previously discussed that a low PMI is associated with postoperative complications in CRC patients [21]. Other studies that examined the relationship between anastomotic failure and sarcopenia also showed that decreased psoas density, a marker of sarcopenia, is correlated with AL among patients who underwent colorectal resection [22, 23], and another report showed that morphological changes of the psoas muscle are associated with overall postoperative complications [24]. However, the patients enrolled in these studies included colon cancer patients who had undergone colonic resection, and no study has examined the relationship between sarcopenia and AL only in male rectal cancer patients who required lower anastomosis without a DS. Thus, it remains controversial whether skeletal muscle mass impacts on AL in rectal cancer patients.
Although AL can occur in cachexic patients with malnutrition, as previously reported, we have frequently experienced anastomotic failure of the colo-procto anastomosis in patients with a good physique such as Judo players. These experiences are inconsistent with previous reports demonstrating that AL was likely to occur in patients with sarcopenia. Male rectal cancer patients with a high BMI and a narrow pelvis are a high-risk group for AL [25]. To develop a novel prediction model for AL, we investigated the impact of the psoas muscle mass on AL in male rectal cancer patients.
This retrospective study was approved by the Ethics Committee of Kitakyushu Municipal Medical Center (No. 201802060). The informed consent requirement was waived based on the nature of this retrospective study, in which patient data was kept confidential. All procedures were conformed to the ethical guideline by the Japanese Government and the Declaration of Helsinki.
Male patients with rectal cancer including rectosigmoid colon who underwent colorectal surgery with low anastomosis below the peritoneal reflection and were free of a DS at our institution from January 2008 until March 2018, were enrolled in this study. All the surgical procedures performed in this study included low anterior resection, super low anterior resection, and intersphincteric anastomosis, and no robotic surgery was performed in this period. We excluded patients with multiple cancers, preoperative bowel obstruction, or colitic cancer and those who had been treated with preoperative adjuvant therapy including neoadjuvant chemoradiotherapy. The medical charts of these patients were retrospectively reviewed.
All CRC patients underwent preoperative abdominal computed tomography (CT) as a routine assessment within 2 months before surgery. Contrast CT was performed except for the patients with contrast agent allergy. Two observers (YM and MS), trained in the identification of the third lumbar vertebrae and in the measurement of the psoas area on CT scan by consultant radiologists, manually traced the bilateral psoas muscle at the intervertebral discs between the third and fourth lumbar vertebrae (L3/L4) and automatically measured the cross-sectional area of the psoas muscle using SYNAPSE VINCENT software (Fujifilm Medical Co., Ltd., Tokyo, Japan). The PMI was calculated as follows according to a previous report [26]: PMI= cross-sectional area of bilateral psoas muscle (cm2)/height2 (m2). The mean PMI value determined by 2 observers was applied in this study as PMI of each patient, as previously reported [27]. Interobserver correlation was assessed to validate whether measurement errors existed.
We performed lymph node dissection according to the Japanese Society for Cancer of the Colon and Rectum guidelines 2019 for the treatment of CRC [28]. Radical surgery with D3 lymph node dissection was performed for locally advanced cancer invading beyond the muscularis propria. The standard concepts of D3 procedure for lower rectal cancer, which is located distal to the peritoneal reflection, include ligation of the inferior mesenteric artery (IMA) at its root and complete mesorectal excision and lateral pelvic lymph node dissection. For early rectal cancer when the tumor was confined above the muscularis propria, we performed D2 lymph node dissection, which included ligation of the superior rectal artery, which is a primary branch of the IMA. For older and high-risk rectal cancer patients, we sometimes performed D3 lymph node dissection with preservation of the left colic artery.
Preoperative variables included sex, age, tumor location, BMI, prognostic nutritional index (PNI), PMI, comorbidity, preoperative serum carcinoembryonic antigen (CEA) level, and carbohydrate antigen (CA) 19-9 level. The intra- and postoperative variables included surgical approach (open or laparoscopic), estimated blood loss volume, surgery duration, left colic artery preservation, usage count of linear stapler for rectal resection, anastomotic site, morbidity, and mortality. Histological variables were tumor histological grade, T stage, and N stage. Tumor location was defined as rectosigmoid and upper and lower rectum. Rectosigmoid colon is defined as the colon from promontory to lower border of second sacrum. Upper rectum is defined as the rectum from lower border of second sacrum to peritoneal reflection. Lower rectum is defined as the rectum from peritoneal reflection to anorectal junction. Tumor differentiation was defined as well differentiated, moderately differentiated, poorly differentiated, and mucinous carcinoma. BMI was divided into high and low BMI groups by the cut-off value of 25.0 kg/m2, which was reported as the cut-off line of obesity in the Asian population [29]. PNI was calculated by the following formula, as proposed by Onodera et al. [30]: PNI= 10× serum albumin level (g/dL)+0.05× total lymphocyte count (mm3). PNI was also divided into 2 groups according to a cut-off value of 40, as previously reported [31]. The upper limit of normal at our institute was defined as a cut-off line of tumor markers (CEA and CA19-9).
Postoperative complications were evaluated by the ClavienDindo (CD) classification [32]. In this study, complications greater than grade II were defined as postoperative complications including AL within 30 days after surgery or before discharge. AL was defined as stool discharge from the intraabdominal drain, positive bacterial culture from the intraabdominal drain, radiological evidence of peritonitis or intraabdominal abscess by CT scan, or enema with rectal contrast. Postoperative mortality indicated death within 30 days after surgery or in-hospital death.
All statistical analyses were performed using JMP 15 software (SAS Institute Inc., Cary, NC, USA). Interobserver correlation of CT measurement was determined by Pearson correlation coefficient [27]. The optimal cut-off value of PMI levels was determined by plotting the area under the receiver operating characteristic (ROC) curve (AUC) and calculating the sensitivity and specificity. The correlation between the 2 variables was analyzed using the chi-square test or Fisher exact test, where appropriate. Continuous variables were expressed as the median, and the range was assessed by the Mann-Whitney U-test. Multiple logistic regression was performed to assess the contribution of significant independent variables in predicting AL using the Cox proportional hazard model. In multivariate analyses, all variables were included in a multivariate backward stepwise logistic regression model to identify significant risk factors for AL, and the variables finally included were PNI, PMI, surgical approach, left colic artery preservation, super low anterior resection, and pathological T stage. Statistical significance was defined as P< 0.05.
Table 1 details the patients’ characteristics. Among all 333 patients with lower anastomosis who underwent sphincter-preserving surgery for rectal cancer including rectosigmoid colon without a DS, preoperative adjuvant therapy, and preoperative bowel obstruction, 197 male patients were enrolled in this study. Fifty-eight male rectal cancer patients with a DS, 136 female patients without a DS, and 28 female patients with a DS are also shown in Supplementary Table 1 as a reference. In the group of male rectal cancer patients without a DS, the mean age of the patients at surgery was 64.3 years (range, 36–81 years). Tumor location was as follows: rectosigmoid (n= 53), upper rectum (n= 65), and lower rectum (n= 79). Laparoscopic surgery was performed in 156 of 197 patients (79.2%). No robotic surgery was performed in this period. CRC staging was as follows: stage 0 (n= 3), stage I (n= 60), stage II (n= 42), stage III (n= 67), and stage IV (n= 25). Median surgery time was 225 minutes (range, 124–588 minutes) and estimated blood loss was 20 mL (range, 1–11,010 mL). Super low anterior resection (distance between anastomotic site and anal verge, < 3 cm) was performed in 60 of 197 patients (30.5%). AL occurred in 45 of 197 patients (22.8%). CD classification was as follows: grade II (n= 14), grade IIIa (n= 5), and grade IIIb (n= 26). No postoperative mortality was found.
We measured the cross-sectional area of the bilateral psoas muscle at the L3/L4 level. PMI was calculated as psoas muscle area in square centimeters divided by height in meters squared. Four examples of CT imaging are shown in Fig. 1. Psoas muscle area was manually traced and automatically measured.
The interobserver correlation of PMI values measured by 2 observers was tested using Pearson correlation coefficient and demonstrated a significant correlation between the 2 observers (r2= 0.973; 95% confidence interval [CI], 0.982–0.989; P< 0.001) (Supplementary Fig. 1). Average PMI value between 2 observers was defined as the personal PMI value, and the median PMI value of the patients was 734.5 (range, 304.4–1,080.1).
We divided the cohort into 2 groups: AL with CD grade of ≥ II (AL+) or not (AL−). Fig. 2 demonstrates that PMI was significantly higher in patients with AL+ than that in AL− patients (median PMI, 856.6 and 713.9, respectively; P< 0.0001). To determine the optimal cut-off value of PMI for AL events, we performed ROC curve analysis (Fig. 3). The AUC value was 0.686 and the optimal cut-off value was 812.67 cm2/m2 (sensitivity 60% and specificity 74.3%). Surprisingly, 27 of the 64 patients (42.2%) experienced AL among the cohort of patients with PMI of > 812.
To determine the predictive factors for AL, univariate analysis of the variables potentially associated with AL was performed and demonstrated that AL significantly occurred in the patients with high PMI ( > 812) and super low anterior resection, indicating that high BMI, history of diabetes mellitus, and nutritional score as known predictive factors for AL did not have a significant difference (Table 2). Multivariate analysis also revealed that high PMI (risk ratio [RR], 3.933; P< 0.001; 95% CI, 1.917–8.070) and super low anastomosis (RR, 2.792; P = 0.015; 95% CI, 1.221–6.384) were independent predictive factors for AL. In total, there were 271 male rectal cancer patients, including those with a DS, preoperative bowel obstruction, and preoperative adjuvant therapy. We reexamined the relationship between AL occurrence and PMI value in the total cohort, which revealed that there was also a significant correlation between PMI value and AL occurrence (P= 0.012). These results suggested that preoperative PMI measurement enabled prediction of the high-risk group for AL among male rectal cancer patients. We should construct a DS in rectal surgery for male patients with a high PMI above the cut-off line.
This study showed that male rectal cancer patients with a large psoas muscle mass who underwent lower anastomosis had more occurrences of postoperative AL than those with smaller psoas muscle masses. We performed abdominal CT for almost all rectal cancer patients; thus, we can easily measure the preoperative PMI of rectal cancer patients. To avoid measurement errors by manual tracing, the mean value measured by 2 observers was adopted as the PMI value in this study, and interobserver correlations between 2 observers were researched by Pearson correlation coefficient, indicating a significant correlation. Previous studies showed that decreased psoas muscle mass, which is a parameter of sarcopenia, increases postoperative complications and is associated with short- and long-term prognostic indicators in colorectal surgery [19, 21, 23, 24]. Our study revealed that high PMI is associated with AL only in male rectal cancer patients undergoing lower anastomosis, and there is a contradiction between our results and previous reports; hence, further discussion is required. The reasons why the probabilities of AL became higher with increased psoas muscle mass may be decreased anatomical space and increased physical force in the anastomotic site due to the large mass of the psoas muscle.
First, large psoas muscle mass can cause the pelvic cavity to be severely narrow due to the reduced anatomical space that is usually already narrow in men, and surgical procedures in the pelvis are extremely difficult, especially total mesorectal excision (TME). In fact, a narrow pelvis is a risk factor of AL [33]. In this study, the PMI was not a risk factor for AL in female patients. Large psoas muscle mass may not influence the pelvic cavity in women because of their relatively wide pelvis, though it can be of influence in men due to their narrow pelvis and large psoas muscle mass compared with women. Previously, Iqbal et al. [34] reported that the extent of TME difficulty depends on a narrower pelvis and a smaller pelvic volume. Large psoas muscle mass makes pelvic dissection difficult due to the narrow pelvis. Patients with a high BMI, another AL predictive factor that increases the difficulty of dissecting the pelvis, also have a narrow pelvis [12, 13]. Novel technologies (robotic surgery) and new approaches (transanal TME) could be of extreme value in overcoming the constraints resulting from a narrow rigid pelvis that are often encountered during conventional rectal surgery [35, 36]. To avoid AL, we should introduce robotic surgery and transanal TME for male patients with a high PMI undergoing rectal surgery.
Second, large psoas muscle mass increases the physical force at the anastomotic site. Previous studies demonstrated that psoas muscle mass is a good indicator of whole-body muscle mass [37], and psoas muscle mass index can be an alternative method of evaluating muscle mass in patients with gastrointestinal and chronic liver disease [38]. Strain pressure in defecation is derived from the abdominal muscle and diaphragm [39]. Thus, we speculated that strain pressure reflects the psoas muscle mass. In this study, the median PMI value was 732 (range, 304.4–1,080.1) in men and 499.2 (range, 239.5–1,033.6) in women, indicating that the psoas muscle of female patients tends not to be large because it is smaller than that of men. In women, high PMI was not risk factor for AL; however, the rate of AL was relatively high (6 of 11, 54.5%), especially in female patients with a high psoas muscle mass (PMI> 700) compared with others (16 of 125, 12.8%), suggesting that the psoas muscle mass can influence AL even in female patients. Women have inherently wide pelvises and a large amount of anatomical space available for surgical procedures compared with men, and there seems to be no issues with female patients undergoing pelvic dissection even if large psoas muscle mass is evident. It is suggested that the reason for high PMI having a negative impact on the anastomotic site is not so much an anatomical issue as a result of the straining abdominal pressure of whole-body skeletal muscle. These results suggest that AL is caused by intraluminal pressure on the anastomotic site caused by strong inner skeletal muscle.
A limitation of our study was that this single institutional retrospective observational analysis of lower rectal cancer patients omitted patients who performed DS, who had received preoperative adjuvant therapy, or who had experienced preoperative bowel obstruction, thus resulting in a strong selection bias and unknown confounding factors. In fact, high BMI is a known predictive marker for AL, but it was not a predictive factor for AL in this study. We excluded patients with a DS because it can be difficult to determine whether AL has occurred because there are fewer AL symptoms in patients with a DS; thus, in the other studies, the proportion of patients with AL can be underestimated. In patients with a high BMI, especially over 30 kg/m2, and malnourished patients with a low BMI (< 18 kg/m2) and low serum albumin, we tend to construct a DS; thus, these patients were excluded from this study. The median BMI in this study was 22.6. In a 2018 national health and nutrition survey, the average BMI of Japanese males was 23.7 [40], higher than that in this study. We measured muscle mass in 2D CT imaging by manual tracing; however, three-dimensional image measurement may ensure more accurate results. In addition, ROC curve analysis showed that the AUC value was not very high, and the cut-off value was not extremely precise (sensitivity of 60% and specificity of 74.3%). The AL rate was slightly high compared with previous reports because the subject of this research was a high-risk group (male patients who had undergone sphincter-preserving surgery without a DS). Among the participants in the study, the patients refusing DS in spite of physicians’ recommendation were included in this study. This study also included the patients with CRC located in rectosigmoid colon and upper rectum, which anastomotic rate is relatively low in general, whereas these patients in this study required longer distal margin and lower anastomosis below the peritoneal reflection due to narrow view and manipulative issue such as bulky mass and comorbid inflammation. All the participants in this study performed lower anastomosis below the peritoneal reflection.
In this study, we demonstrated that large psoas muscle mass is a predictive marker for AL in male rectal cancer patients. We recommend measuring psoas muscle area and preoperatively evaluating PMI, and constructing a DS when the PMI is higher than the cut-off value. We should also elucidate the mechanism by which psoas muscle mass affects AL in male rectal cancer patients in a large-scale multi-centered prospective study.
SUPPLEMENTARY MATERIALS
Supplementary materials for this study are presented online (available at https://doi.org/10.3393/ac.2022.00122.0017).
Supplementary Table 1.
Comparison of clinicopathological factors among the 4 groups of rectal cancer patients
Supplementary Fig. 2.
The interobserver correlation of psoas muscle index values measured by 2 observers was tested using Pearson correlation coefficient and demonstrated a significant correlation between the 2 observers (r2= 0.973; 95% CI, 0.982–0.989; P < 0.001).
Fig. 1.
Four examples of computed tomography (CT) imaging showing manual tracing and automatic measurement of the cross-sectional area of the psoas muscle. CT images refer to patients with (A, B) high psoas muscle index (PMI) and (C, D) low PMI.

Fig. 2.
Median psoas muscle index (PMI) was significantly higher in patients with anastomotic leakage (AL+; Clavien-Dindo grade ≥ II) than in AL− patients (856.6 vs. 713.9, P < 0.001).

Fig. 3.
Receiver operating characteristic curve analysis showed that the area under the curve (AUC) value was 0.686 and the optimal cut-off value was 812.67 cm2/m2 (sensitivity of 60% and specificity of 74.3%).
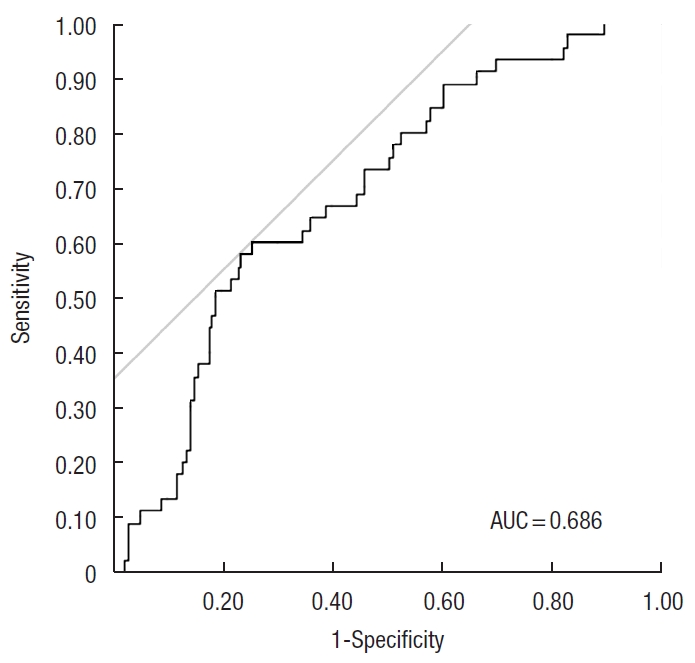
Table 1.
Clinicopathological factors in male rectal cancer patients without ileostomy
Table 2.
Univariate and multivariate analysis of predictive factors for AL in male rectal cancer patients without ileostomy
REFERENCES
2. Cancer Information Service, National Cancer Center Japan. Cancer registry and statistics [Internet]. Tokyo, National Cancer Center Japan; 2017 [cited 2022 Jan 24]. Available from: http://ganjoho.jp/reg_stat/statistics/dl/index.html#incidence
3. Inomata M, Shiroshita H, Uchida H, Bandoh T, Akira S, Yamaguchi S, et al. Current status of endoscopic surgery in Japan: the 14th National Survey of Endoscopic Surgery by the Japan Society for Endoscopic Surgery. Asian J Endosc Surg 2020;13:7–18.



4. Wang S, Liu J, Wang S, Zhao H, Ge S, Wang W. Adverse effects of anastomotic leakage on local recurrence and survival after curative anterior resection for rectal cancer: a systematic review and meta-analysis. World J Surg 2017;41:277–84.




5. Kulu Y, Tarantio I, Warschkow R, Kny S, Schneider M, Schmied BM, et al. Anastomotic leakage is associated with impaired overall and disease-free survival after curative rectal cancer resection: a propensity score analysis. Ann Surg Oncol 2015;22:2059–67.



6. Mirnezami A, Mirnezami R, Chandrakumaran K, Sasapu K, Sagar P, Finan P. Increased local recurrence and reduced survival from colorectal cancer following anastomotic leak: systematic review and meta-analysis. Ann Surg 2011;253:890–9.

7. Hüser N, Michalski CW, Erkan M, Schuster T, Rosenberg R, Kleeff J, et al. Systematic review and meta-analysis of the role of defunctioning stoma in low rectal cancer surgery. Ann Surg 2008;248:52–60.


8. Neuman HB, Patil S, Fuzesi S, Wong WD, Weiser MR, Guillem JG, et al. Impact of a temporary stoma on the quality of life of rectal cancer patients undergoing treatment. Ann Surg Oncol 2011;18:1397–403.



9. Watanabe J, Ishibe A, Suwa Y, Suwa H, Ota M, Kunisaki C, et al. Indocyanine green fluorescence imaging to reduce the risk of anastomotic leakage in laparoscopic low anterior resection for rectal cancer: a propensity score-matched cohort study. Surg Endosc 2020;34:202–8.



10. Blanco-Colino R, Espin-Basany E. Intraoperative use of ICG fluorescence imaging to reduce the risk of anastomotic leakage in colorectal surgery: a systematic review and meta-analysis. Tech Coloproctol 2018;22:15–23.



11. Ito T, Obama K, Sato T, Matsuo K, Inoue H, Kubota K, et al. Usefulness of transanal tube placement for prevention of anastomotic leakage following laparoscopic low anterior resection. Asian J Endosc Surg 2017;10:17–22.



12. Sparreboom CL, van Groningen JT, Lingsma HF, Wouters M, Menon AG, Kleinrensink GJ, et al. Different risk factors for early and late colorectal anastomotic leakage in a nationwide audit. Dis Colon Rectum 2018;61:1258–66.


13. Qu H, Liu Y, Bi DS. Clinical risk factors for anastomotic leakage after laparoscopic anterior resection for rectal cancer: a systematic review and meta-analysis. Surg Endosc 2015;29:3608–17.



14. Paireder M, Asari R, Kristo I, Rieder E, Tamandl D, Ba-Ssalamah A, et al. Impact of sarcopenia on outcome in patients with esophageal resection following neoadjuvant chemotherapy for esophageal cancer. Eur J Surg Oncol 2017;43:478–84.


15. Kawamura T, Makuuchi R, Tokunaga M, Tanizawa Y, Bando E, Yasui H, et al. Long-term outcomes of gastric cancer patients with preoperative sarcopenia. Ann Surg Oncol 2018;25:1625–32.



16. Mintziras I, Miligkos M, Wächter S, Manoharan J, Maurer E, Bartsch DK. Sarcopenia and sarcopenic obesity are significantly associated with poorer overall survival in patients with pancreatic cancer: Systematic review and meta-analysis. Int J Surg 2018;59:19–26.


17. Elliott JA, Doyle SL, Murphy CF, King S, Guinan EM, Beddy P, et al. Sarcopenia: prevalence, and impact on operative and oncologic outcomes in the multimodal management of locally advanced esophageal cancer. Ann Surg 2017;266:822–30.

18. Voron T, Tselikas L, Pietrasz D, Pigneur F, Laurent A, Compagnon P, et al. Sarcopenia impacts on short- and long-term results of hepatectomy for hepatocellular carcinoma. Ann Surg 2015;261:1173–83.


19. Sun G, Li Y, Peng Y, Lu D, Zhang F, Cui X, et al. Can sarcopenia be a predictor of prognosis for patients with non-metastatic colorectal cancer? A systematic review and meta-analysis. Int J Colorectal Dis 2018;33:1419–27.



20. Prado CM, Lieffers JR, McCargar LJ, Reiman T, Sawyer MB, Martin L, et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol 2008;9:629–35.


21. Nakanishi R, Oki E, Sasaki S, Hirose K, Jogo T, Edahiro K, et al. Sarcopenia is an independent predictor of complications after colorectal cancer surgery. Surg Today 2018;48:151–7.



22. Herrod P, Boyd-Carson H, Doleman B, Trotter J, Schlichtemeier S, Sathanapally G, et al. Quick and simple: psoas density measurement is an independent predictor of anastomotic leak and other complications after colorectal resection. Tech Coloproctol 2019;23:129–34.




23. Margadant CC, Bruns ER, Sloothaak DA, van Duijvendijk P, van Raamt AF, van der Zaag HJ, et al. Lower muscle density is associated with major postoperative complications in older patients after surgery for colorectal cancer. Eur J Surg Oncol 2016;42:1654–9.


24. Hanaoka M, Yasuno M, Ishiguro M, Yamauchi S, Kikuchi A, Tokura M, et al. Morphologic change of the psoas muscle as a surrogate marker of sarcopenia and predictor of complications after colorectal cancer surgery. Int J Colorectal Dis 2017;32:847–56.



25. McDermott FD, Heeney A, Kelly ME, Steele RJ, Carlson GL, Winter DC. Systematic review of preoperative, intraoperative and postoperative risk factors for colorectal anastomotic leaks. Br J Surg 2015;102:462–79.



26. van Vugt JL, Levolger S, de Bruin RW, van Rosmalen J, Metselaar HJ, IJzermans JN. Systematic review and meta-analysis of the impact of computed tomography-assessed skeletal muscle mass on outcome in patients awaiting or undergoing liver transplantation. Am J Transplant 2016;16:2277–92.



27. Richards SJ, Senadeera SC, Frizelle FA. Sarcopenia, as assessed by psoas cross-sectional area, is predictive of adverse postoperative outcomes in patients undergoing colorectal cancer surgery. Dis Colon Rectum 2020;63:807–15.


28. Hashiguchi Y, Muro K, Saito Y, Ito Y, Ajioka Y, Hamaguchi T, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol 2020;25:1–42.




29. WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 2004;363:157–63.


30. Onodera T, Goseki N, Kosaki G. Prognostic nutritional index in gastrointestinal surgery of malnourished cancer patients. Nihon Geka Gakkai Zasshi 1984;85:1001–5.

31. Miyakita H, Sadahiro S, Saito G, Okada K, Tanaka A, Suzuki T. Risk scores as useful predictors of perioperative complications in patients with rectal cancer who received radical surgery. Int J Clin Oncol 2017;22:324–31.




32. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205–13.


33. Tsuruta A, Tashiro J, Ishii T, Oka Y, Suzuki A, Kondo H, et al. Prediction of anastomotic leakage after laparoscopic low anterior resection in male rectal cancer by pelvic measurement in magnetic resonance imaging. Surg Laparosc Endosc Percutan Tech 2017;27:54–9.



34. Iqbal A, Khan A, George TJ, Tan S, Qiu P, Yang K, et al. Objective preoperative parameters predict difficult pelvic dissections and clinical outcomes. J Surg Res 2018;232:15–25.


35. Aselmann H, Kersebaum JN, Bernsmeier A, Beckmann JH, Möller T, Egberts JH, et al. Robotic-assisted total mesorectal excision (TME) for rectal cancer results in a significantly higher quality of TME specimen compared to the laparoscopic approach-report of a single-center experience. Int J Colorectal Dis 2018;33:1575–81.



36. Atallah S, Martin-Perez B, Albert M, deBeche-Adams T, Nassif G, Hunter L, et al. Transanal minimally invasive surgery for total mesorectal excision (TAMIS-TME): results and experience with the first 20 patients undergoing curative-intent rectal cancer surgery at a single institution. Tech Coloproctol 2014;18:473–80.



37. Morrell GR, Ikizler TA, Chen X, Heilbrun ME, Wei G, Boucher R, et al. Psoas muscle cross-sectional area as a measure of wholebody lean muscle mass in maintenance hemodialysis patients. J Ren Nutr 2016;26:258–64.



38. Takada H, Amemiya F, Yasumura T, Yoda H, Okuwaki T, Tanaka K, et al. Utility of the simplified measurements of muscle mass in patients with gastrointestinal and chronic liver diseases. Sci Rep 2020;10:10795.




40. Ministry of Health, Labour and Welfare. Outline of results from 2018 National Health and Nutrition Survey in Japan [Internet]. Tokyo, Ministry of Health, Labour and Welfare; 2018 [cited 2022 Jan 24]. Available from: https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/kenkou_iryou/kenkou/eiyou/h30-houkoku_00001.html
- TOOLS