Impact of consolidation chemotherapy in poor responders to neoadjuvant radiation therapy: magnetic resonance imaging–based clinical-radiological correlation in high-risk rectal cancers
Article information
Abstract
Purpose
The current study was conducted to examine the role of consolidation chemotherapy after neoadjuvant radiation therapy (NART) in decreasing the involvement of the mesorectal fascia (MRF) in high-risk locally advanced rectal cancers (LARCs).
Methods
In total, 46 patients who received consolidation chemotherapy after NART due to persistent MRF involvement were identified from a database. A team of 2 radiologists, blinded to the clinical data, studied sequential magnetic resonance imaging (MRI) scans to assess the tumor response and then predict a surgical plan. This prediction was then correlated with the actual procedure conducted as well as histopathological details to assess the impact of consolidation chemotherapy.
Results
The comparison of MRI-based parameters of sequential images showed significant downstaging of T2 signal intensity, tumor height, MRF involvement, diffusion restriction, and N category between sequential MRIs (P<0.05). However, clinically relevant downstaging (standardized mean difference, >0.3) was observed for only T2 signal intensity and diffusion restriction on diffusion-weighted imaging. No clinically relevant changes occurred in the remaining parameters; thus, no change was noted in the extent of surgery predicted by MRI. Weak agreement (Cohen κ coefficient, 0.375) and correlation (Spearman rank coefficient, 0.231) were found between MRI-predicted surgery and the actual procedure performed. The comparison of MRI-based and pathological tumor response grading also showed a poor correlation.
Conclusion
Evidence is lacking regarding the use of consolidation chemotherapy in reducing MRF involvement in LARCs. The benefit of additional chemotherapy after NART in decreasing the extent of planned surgery by reducing margin involvement requires prospective research.
INTRODUCTION
In the current climate of total neoadjuvant therapy (TNT) for locally advanced rectal cancers (LARCs), the preoperative use of chemotherapy has gained acceptance for a wide spectrum of reasons. Two recent trials, RAPIDO (Rectal Cancer and Preoperative Induction Therapy Followed by Dedicated Operation) [1] and UNICANCER-PRODIGE 23 [2], have been instrumental in paving the way for a TNT approach in LARC treatment. Both trials showed improvements in distant metastasis-free survival and pathological complete response rates. Despite this, no improvement was observed in the local control rate or in overall survival. Similarly, the OPRA (Organ Preservation of Rectal Adenocarcinoma) trial [3] provided a head-to-head comparison between the 2 TNT approaches—induction chemotherapy and consolidation chemotherapy—and showed superior organ preservation with a consolidation chemotherapy approach. However, the overall survival rate was similar to historical control rates. Indirect evidence indicates that consolidation chemotherapy works best in patients with good responses to neoadjuvant radiation therapy (NART; “good responders”). The role of consolidation chemotherapy in patients with poor response to radiation therapy (“poor responders”) is uncertain [4]. The popular use of consolidation chemotherapy in poor responders, especially those with persistent involvement of mesorectal fascia (MRF) after NART, is not evidence-based. The real-time benefit of additional chemotherapy in reducing MRF involvement, and thereby decreasing the extent of surgery, has not been assessed. The current study, focusing on clinical-radiological correlation, was conducted to assess the impact of consolidation chemotherapy in downstaging the MRF involvement in LARCs with persistent involvement of MRF after NART.
The primary objective was to assess the impact of consolidation chemotherapy on MRF involvement in patients who had received NART. The secondary objective was to correlate the radiological response after consolidation chemotherapy, as shown on magnetic resonance imaging (MRI)-based local disease imaging, with the actual procedure performed.
METHODS
Ethics statement
This study was approved by the Institutional Review Board of Tata Memorial Centre (No. 1478). The requirement for informed consent was waived due to the retrospective nature of the study. The study was conducted in accordance with the published ethical standards of the Declaration of Helsinki.
Study design
This was a retrospective observational study. The departmental database in the colorectal division of Tata Memorial Centre (Mumbai, India) was screened from January 2018 to December 2019 to identify patients receiving consolidation chemotherapy with the intent to decrease MRF involvement during the treatment course. Demographic, diagnostic, and treatment-related variables were retrieved from electronic medical records maintained on an online platform.
Participants
The inclusion criteria were the following: patients with rectal adenocarcinoma between 18 and 75 years of age; patients who had received consolidation chemotherapy after NART in view of MRF involvement; and patients for whom all of the response assessment MRI scans were available for radiological review. The exclusion criteria were the following: patients for whom serial MRI scans were not available for review by the radiology team and patients who had received consolidation chemotherapy for reasons other than MRF involvement.
Disease management protocol
During the study period, rectal adenocarcinoma cases were staged at presentation using pelvic MRI (baseline MRI, MRI-1) and a contrast-enhanced computed tomography scan of the abdomen and thorax. For patients with locally advanced cancers of the mid-and low-rectum, neoadjuvant long-course chemoradiation therapy was planned with 50.4-Gy external beam radiation in 28 fractions and concurrent 5-fluorouracil or capecitabine. The use of short-course radiation therapy was limited to either the metastatic setting (to facilitate introduction of chemotherapy early in the treatment paradigm) or cases requiring early surgery. Six weeks after the last dose of radiation, patients underwent response assessment involving pelvic MRI (MRI after neoadjuvant radiation therapy, MRI-2) and clinical examination. Restaging for distant metastases was performed in cases of progressive disease. For patients with persistent involvement of the MRF by the primary disease on MRI-2, consolidation chemotherapy (4 cycles of capecitabine/oxaliplatin or 5-fluorouracil/oxaliplatin) was planned to reduce the MRF involvement. Pelvic MRI for local response assessment was repeated (MRI after consolidation chemotherapy, MRI-3). Patients then underwent surgery in the form of total mesorectal excision (TME), beyond TME (b-TME), or extended TME (e-TME) after a tumor board discussion. Surgery was classified as within or beyond the TME plane per the consensus statement by the Beyond TME Collaborative [5]. After recovery from surgery, patients were advised regarding adjuvant chemotherapy.
Data gathering and review
Serial MRI scans of the included patients were retrieved and reviewed by a team of 2 dedicated radiologists from the colorectal disease management group. The radiologists were blinded to the clinical data. Patients were only included for whom at least MRI-2 and MRI-3 records were available for radiology review (Fig. 1).
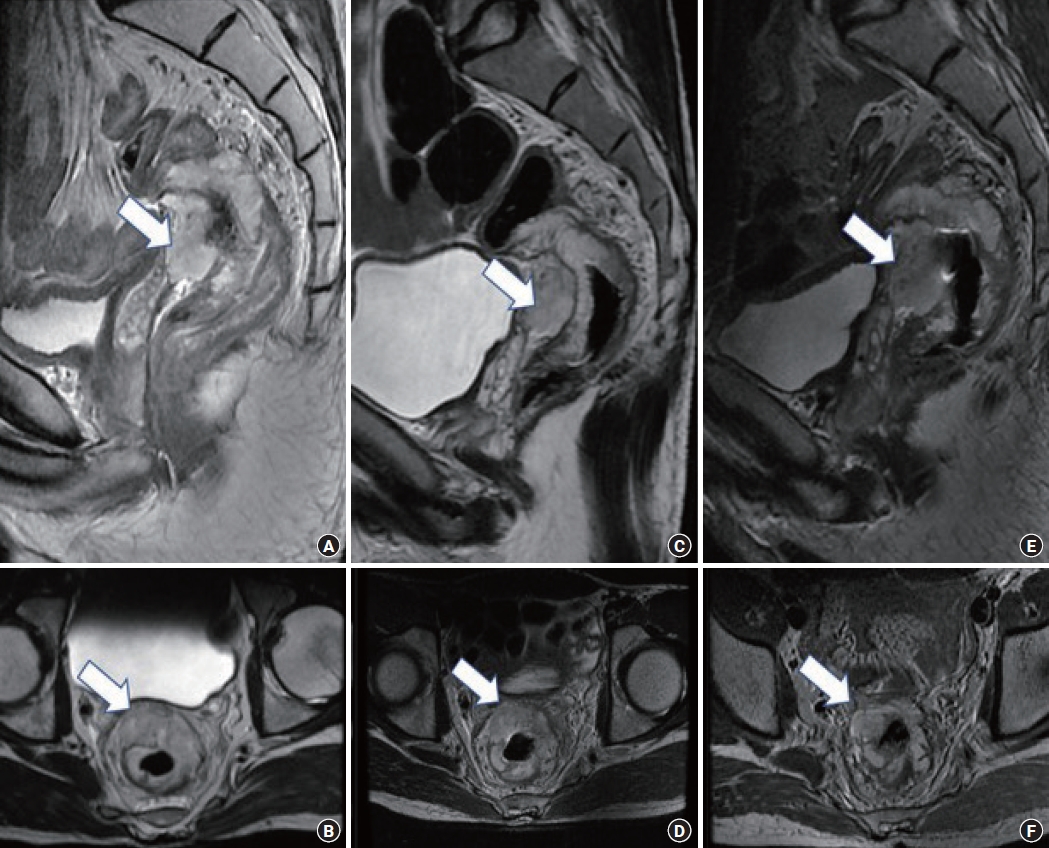
Sequential pelvic magnetic resonance imaging (MRI) comparisons assessing the response to neoadjuvant radiation therapy and additional consolidation chemotherapy. (A, B) Baseline MRI (MRI-1) showed a mucinous tumor (arrows) in the mid-rectum abutting the seminal vesicle. (C, D) MRI after neoadjuvant radiation therapy (MRI-2) showed no response to therapy. (E, F) MRI after consolidation chemotherapy (MRI-3) showed a mild increase in the bulk of mucin (arrows) abutting the seminal vesicle.
Working definitions
Radiologically, the MRF was considered involved if the farthest distance of the disease from the margin was less than 1 mm. The MRI-based response assessment included the evaluation of diffusion restriction on diffusion-weighted imaging (DWI) and signal intensity on T2 sequences along with volumetric parameters. Changes in the extramesorectal nodes, extramural vascular invasion (EMVI), and the site of MRF involvement were noted. The MRI-based tumor regression grade (MR-TRG) was also documented [6]. Any overall increase or decrease in the disease was documented in consideration of all parameters together according to radiologist discretion. The various radiological MRI-based parameters were considered together as potential predictors of clinically relevant changes in MRI parameters. The clinical relevance of the MRI parameters was determined by the radiology team based on whether any change was indicated in the extent of predicted surgery. The change in individual parameters between sequential MRI scans was analyzed statistically as detailed below.
Correspondingly, the pathological circumferential resection margin (CRM) was considered to be positive if a tumor or tumorcell–bearing mucin was found within 1 mm of the CRM. The Mandard classification system was used to determine the pathological TRG (P-TRG) [7].
Statistical analysis
The data were analyzed using IBM SPSS ver. 26.0 (IBM Corp). The Wilcoxon signed rank test was used to compare continuous variables. The McNemar-Bowker marginal test for homogeneity was used to compare ordinal variables, and the McNemar test was used to compare nominal variables. A standardized mean difference of more than 0.3 was considered to denote a clinically relevant change in variables. Correlations were measured using the Spearman rank coefficient, while the Cohen κ coefficient was used for tests of agreement.
RESULTS
Overall, 46 patients with LARCs with involvement of MRF on MRI-1 were screened. After NART, 4 patients exhibited free MRF on MRI-2, while the remaining 42 patients had persistent MRF involvement. The latter patients were then offered consolidation chemotherapy to reduce the involvement of the MRF. On MRI-3, 12 patients had downstaging of MRF involvement, while the remaining 30 had persistent involvement of the MRF. A cohort diagram is presented in Fig. 2, and the disease and treatment characteristics are shown in Table 1.
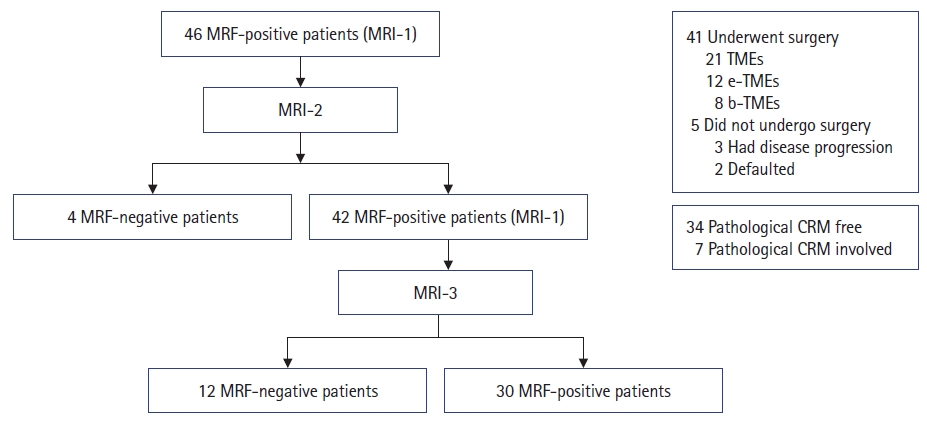
Cohort diagram. MRF, mesorectal fascia; MRI, magnetic resonance imaging; MRI-1, baseline MRI; MRI-2, MRI after neoadjuvant radiation therapy; MRI-3, MRI after consolidation chemotherapy; TME, total mesorectal excision; e-TME, extended TME; b-TME, beyond TME; CRM, circumferential resection margin.
Comparison of MRI-2 and MRI-3
MRI parameters studied and compared between MRI-2 and MRI-3 included T2 signal intensity, tumor length, MRF involvement, EMVI, restriction on DWI, T category, N category, extramesorectal lateral pelvic nodes, and involvement of the sphincter-levator muscle complex
The authors observed significant downstaging of T2 signal intensity, tumor length, involvement of MRF, diffusion restriction, and N category between MRI-2 and MRI-3 (P<0.05 on tests of symmetry). No significant change was observed for T category, extramesorectal nodes, EMVI, and involvement of the sphincter-levator complex upon serial comparison between MRI-2 and MRI-3 (Table 2).
The serial values of the MRI parameters were examined for any clinically relevant changes, and radiologists reported the MRI-predicted extent of surgery based on the 2 sequential MRIs. Among the morphological parameters, clinically relevant downstaging was observed for only T2 signal intensity and diffusion restriction on DWI (standardized mean difference, >0.3). No clinically relevant change was noted in the remaining parameters, and thus no change was found in MRI-predicted surgery.
Concordance between MRI prediction of surgery and actual surgery performed
Upon analysis of 41 patients who underwent surgery, surgery was performed in the TME plane, e-TME plane, and b-TME plane in 21, 12, and 8 patients, respectively. Five patients underwent no surgery: 3 patients had disease progression, and 2 patients with planned exenteration opted out and defaulted.
The radiologist-predicted extent of surgery based on MRI alone was compared to the actual procedure performed. Agreement testing showed a Cohen κ coefficient of 0.375 (standard error, 0.125; P=0.001), suggesting a weak agreement between the 2 variables. Similarly, correlation testing revealed a Spearman rank coefficient of 0.231 (standard error, 0.164; P=0.146), suggesting a weak correlation (Table 3).
Concordance between MR-TRG and P-TRG
The 5-point MR-TRG system was compared and correlated with the similar P-TRG system to identify any potential association between the 2 methods of assessing tumor response to neoadjuvant treatment. Agreement testing revealed a Cohen kappa coefficient of −0.017 (standard error, −0.082; P=0.822), suggesting disagreement between the methods. Correlation testing revealed a Spearman rank correlation coefficient of 0.205 (standard error, 0.161; P=0.122), suggesting a weak correlation (Table 4).
Regression analysis for factors impacting radiological response
Binomial regression analysis was used to analyze the impact of various baseline disease factors (T category, N category, presence of extramesorectal nodes, quadrant of MRF involvement, histopathological variety, type of radiation therapy, and number of cycles of consolidation chemotherapy) on the radiological response (change in MRI-based parameters after completion of all neoadjuvant treatment) (Table 5). Disease histology significantly impacted the degree of radiological response to neoadjuvant treatment. Twenty patients with poorly differentiated or signet ring cell tumors had an inferior response relative to 26 patients with well or moderately differentiated tumors (hazard ratio, 0.134; 95% confidence interval, 0.026–0.687; P=0.016).
DISCUSSION
In the rapidly evolving treatment paradigm for rectal cancer, survival outcomes are an attribute of the stage at presentation, treatment received, and overall disease biology. The recently published RAPIDO trial [1] and UNICANCER-PRODIGE 23 trial [2] provide evidence for inculcation of a complete chemotherapy schedule in the neoadjuvant setting, reducing the risk of distant metastatic relapse. While the RAPIDO trial [1] involved short-course radiation therapy with 6 cycles of capecitabine and oxaliplatin (CAPOX) or fluorouracil, leucovorin, and oxaliplatin (FOLFOX) for high-risk LARCs, the UNICANCER-PRODIGE 23 trial [2] involved 6 cycles of induction chemotherapy with long-course chemoradiation therapy for all LARCs. The high-risk LARC subgroup included patients with T3 and T4 node-positive cancers as well as MRF involvement, presence of lateral lymph nodes, or EMVI. The results indicated a 7.5% increment in distant metastasis-free survival and a near doubling in the pathological complete response rate. However, no improvement was observed in either overall survival or local disease control. Similarly, the OPRA trial [3] showed improvement in organ preservation with a consolidation chemotherapy-based TNT approach relative to induction chemotherapy-based TNT. Overall survival outcomes of both TNT approaches remained similar to the historical control, likely because consolidation chemotherapy selectively benefits good responders.
Experts are debating the role of adjuvant chemotherapy after preoperative radiation therapy [8]. It possibly seems superfluous to subject patients to additional chemotherapy for a 7.5% increment in distant metastasis-free survival without any overall survival advantage. No trials have reported on the actuarial rates of local downstaging observed with the use of consolidation chemotherapy. Analyzing the results of the STELLAR trial, Negri and Aschele [4] suggested a careful risk profile-based use of consolidation chemotherapy over and above radiation therapy. The role of additional chemotherapy (consolidation chemotherapy) is uncertain and remains to be studied.
In the current study, the authors analyzed the treatment course of high-risk LARCs that were treated with consolidation chemotherapy in view of persistent involvement of MRF following chemoradiation. An MRI-based repeat assessment was done after at least 4 cycles of consolidation chemotherapy. The mean age of the cohort was 38.5 years, and 43.5% of patients had poorly differentiated histology with or without signet ring cells. This patient population is reflective of the population with aggressive cancer increasingly seen in Asia [9].
MRI has progressively made its way into the diagnostic and treatment algorithm of LARCs. In 2006, the MERCURY Study Group described MRI as the most appropriate tool to predict the surgical planes for rectal cancer, with superior local control rates [10]. Kulkarni et al. [11] showed that reassessment MRI scans have good specificity and high negative predictive value for predicting CRM and tumor response; however, they have low sensitivity. Adding to the initial experience of the MERCURY Study Group, Patel et al. [6] demonstrated the utility of restaging MRI to differentiate good and poor responders to standard chemoradiation and tailor further neoadjuvant treatment accordingly prior to surgical treatment. The MR-TRG system has been shown to help differentiate the response to chemoradiation [7, 12]. With the MERCURY II Study, Battersby et al. [13] showed the predictive capability of MRI in guiding the plane of surgery for high-risk rectal cancers. DWI sequences have gained acceptance in clinical application for response assessment after neoadjuvant radiation over the last decade [14].
The current report describes changes in various morphometric MRI parameters during the neoadjuvant treatment course of LARCs. Use of consolidation chemotherapy after NART led to downstaging in several parameters, including T2 signal intensity, tumor length, MRF involvement, diffusion restriction, and N category between sequential response assessment MRI scans. However, clinically relevant downstaging was seen for only T2 signal intensity and diffusion restriction on DWI. Thus, after combining the clinical relevance of various parameters to predict the actual plane of surgery, no change was seen in the MRI-predicted extent of the procedure.
Signet ring cell adenocarcinomas (SRCAs) are known to respond relatively poorly to standard chemoradiation treatment [15, 16]. In this study, poorly differentiated tumors with or without signet ring cell histology showed a poor radiological response to neoadjuvant treatment in comparison to well or moderately differentiated tumors. A previously published report from the present authors’ team emphasized the same [15]. Patients with signet ring cell tumors had inferior response rates to traditional neoadjuvant treatment strategies, with inferior survival relative to patients with non–signet tumors (P<0.001). The superiority of the TNT strategy in SRCA cancers remains to be confirmed. Signet ring cells are currently being tested in organoids for treatment with different systemic agents [17].
The current study revealed a poor correlation between the MRI-predicted extent of surgery and the actual procedure performed. The MRI-predicted tumor response did not correspond with the actual histopathological tumor response. The authors postulate that the utility of MRI for response assessment in this high-risk SRCA cohort requires further testing with a larger sample size. The high-risk SRCA patients could underlie the poor capability of MRI parameters to appropriately predict the tumor response in the current study.
The addition of consolidation chemotherapy has been shown to increase the pathological downstaging of LARCs, resulting in increased pathological complete response rates without any clinically relevant increase in toxicity profile [18–20]. However, the use of consolidation chemotherapy to downstage the MRF involvement and thereby decrease the extent of surgery remains to be proven. In the RAPIDO trial [1], the positive CRM rate was 9% in both the standard and experimental arms. Inadequate MRI-based sequential response assessment in high-risk LARCs has been highlighted as a contributor to the lower-than-expected benefit of the TNT approach used in the RAPIDO trial [21]. The results of MRI-based response assessment for high-risk LARCs in the RAPIDO trial [1] and UNICANCER-PRODIGE 23 trial [2] will further elucidate the real-time effect of consolidation chemotherapy on local downstaging. The TRIGGER study [22] is a feasibility study currently being conducted to stratify the management of LARCs according to MRI-based assessment. In that study, researchers are testing the utility of MR-TRG in evaluating the response to NART and differentiating between good and poor responders.
A strength of the current report is that it is the first study to elucidate the relationship between consolidation chemotherapy and objective changes in MRI parameters, in the subgroup of mucinous tumors. The study involved analysis of a high-risk subgroup for which consolidation chemotherapy is often used for local downstaging without any objective evidence. The study emphasizes the need for a dedicated radiology team to guide the treatment protocol based on timely sequential MRI assessments.
The limitations of this study relate to its retrospective design and limited sample size. LARCs with poor differentiation are known to display inferior response to neoadjuvant treatment, and this could affect the predictive capacity of MRI-based response assessment.
The authors emphasize the need for evidence supporting the use of consolidation chemotherapy for local downstaging to increase the resectability of high-risk rectal cancers. The unwarranted use of consolidation chemotherapy in nonresponders potentially increases the time to surgery without any decrease in the planned extent of surgical resection. This could underlie poor outcomes in the high-risk subgroup of LARCs with MRF involvement at baseline. We suggest that it may be reasonable to increase the extent of surgery after radiation therapy for these poorly responsive tumors as opposed to subjecting patients to further chemotherapy with little real-time benefit.
This was a hypothesis-generating study. Various aspects of the treatment of mucinous rectal cancers, including the responsiveness to chemotherapy, the utility of MRI in response assessment, and appropriate time points for response assessment, require prospective research with a large sample size.
In conclusion, evidence is lacking regarding the use of consolidation chemotherapy in reducing MRF involvement in LARCs. The potential benefit of additional chemotherapy after NART in decreasing the extent of planned surgery by reducing margin involvement requires prospective research with a large sample size to draw practice-changing conclusions. The use of MRI for predicting the extent of surgery (TME, e-TME, or b-TME) after NART in patients with mucinous histology at baseline requires prospective validation.
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Funding
None.
Author contributions
Conceptualization: SP, AS; Data curation: SP, SA, PH; Formal analysis: SP, SA, MK; Methodology: SP, SA, PH; Project administration: ALD, AS; Supervision: AS; Writing–original draft: SP, SA; Writing–review & editing: all authors. All authors read and approved the final manuscript.





