Characteristics of Patients Presented With Metastases During or After Completion of Chemoradiation Therapy for Locally Advanced Rectal Cancer: A Case Series
Article information
Abstract
Purpose
Locally advanced rectal cancer (LARC) is managed by chemoradiotherapy (CRT), followed by surgery. Herein we reported patients with metastases during or after CRT.
Methods
Data of patients with LARC who received CRT from 2008 to 2017 were reviewed. Patients with metastases after CRT were included. Those with metastatic tumors at the initial diagnosis were excluded.
Results
Fourteen patients (1.3%) of 1,092 who received CRT presented with metastases. Magnetic resonance circumferential resection margin (mrCRM) and mesorectal lymph nodes (LNs) were positive in 12 patients (85.7%). Meanwhile, magnetic resonance extramural vascular invasion (mrEMVI) was positive in 10 patients (71.4%). Magnetic resonance tumor regression grade (mrTRG) 4 and mrTRG5 was detected in 5 and 1 patient respectively. Ten patients (71.4%) underwent combined surgery and 3 (21.4%) received palliative chemotherapy.
Conclusion
Patients with metastases after CRT showed a higher rate of positive mrCRM, mrEMVI, mesorectal LNs, and poor tumor response. Further studies with a large number of patients are necessary for better survival outcomes in LARC.
INTRODUCTION
In the past, locally advanced rectal cancers (LARCs) were defined as stage T3 to T4 cancers or those with positive lymph nodes (LNs) according to the American Joint Committee on Cancer staging system [1]. Conversely, some authors consider only T4 as a LARC, regardless of whether it is associated with positive LNs [2]. Currently, the typical definition of LARCs is those which require multidisciplinary management and includes tumors extending beyond the total mesorectal excision (TME) planes that need pelvic exenteration, advanced T stage (≥ T3) tumors or positive mesorectal LNs with or without extramural venous invasion (EMVI) and positive circumferential resection margin (CRM) [3, 4].
According to the National Comprehensive Cancer Network guidelines, there are some treatment strategies for LARC. Moreover, total neoadjuvant therapy has been widely studied around the world. However, neoadjuvant long-course chemoradiotherapy (CRT) over 5 weeks, followed by TME after a gap period of 8 to 10 weeks, and then postoperative chemotherapy was traditionally used in Korea and Western countries [5]. Regarding neoadjuvant chemotherapy, a small dose of capecitabine is used as a radiosensitizer, which is a safe and tolerable drug that increases the rate of sphincter-saving surgery [5, 6]. After the completion of CRT, a waiting period is mandatory for tumor response and/or regression because the rectal cancer response to CRT is time-dependent. Meanwhile, this gap period is used for the evaluation and restaging of the tumor after treatment [7].
Multiple studies have demonstrated the benefits of CRT, including downstaging and downsizing, which in turn enhance the possibility of sphincter preservation surgery [7-9]. Although preoperative CRT has markedly decreased local recurrence with a variety of tumor responses ranging from pathologically complete response to no response or even progression during treatment, its role in the control of distant metastases is considered a matter of concern [9-11].
During the gap period before surgery, we sometimes encounter patients with metastases during or after completion of CRT for whom combined surgery or salvage chemotherapy is conducted but with poor outcomes. Therefore, we aimed to summarize the characteristics and clinical course of patients who had metastasis after CRT.
METHODS
Patient enrollment
The retrospectively collected data of 1,092 patients with LARC who received preoperative CRT at Department of Surgery, Severance Hospital, Yonsei University College of Medicine (Seoul, Korea) from 2008 to 2017 were reviewed. We included patients who developed organ metastases during or after the completion of CRT. Those with metastatic tumors present at initial imaging were excluded. The clinical and radiological characteristics of the patients were reported. The study was approved by the Institutional Review Board of Severance Hospital (No. 4-2019-1347). We got the written informed consent from the patients.
Preoperative assessment and neoadjuvant CRT
Patients were evaluated by clinical examination, abdominopelvic computed tomography (CT) scan, serum carcinoembryonic antigen (CEA) level, chest CT, and pelvic magnetic resonance imaging (MRI). All patients were diagnosed with LARC using MRI. The tumor pathology was determined to be adenocarcinoma by colonoscopic biopsy.
Patients received neoadjuvant CRT (50.4 Gy in 28 fractions) for 5 to 6 weeks with capecitabine as a radiosensitizer. All patients were evaluated by abdominopelvic CT, chest CT, pelvic MRI, and CEA level after an 8- to 10-week gap to evaluate response.
Methods of intervention
Patients diagnosed with metastases in the waiting period after CRT were requested to wait till the multidisciplinary team meeting to receive a final decision regarding their treatment. Those who presented with LARC associated with resectable metastases to the liver and/or lung with or without peritoneal metastases and had no contraindications for surgery underwent combined resection either directly or after upfront chemotherapy (if R0 resection was possible). All decisions were made by a colorectal surgeon as well as a hepatobiliary and/or cardiothoracic surgeon. Contrarily, patients who developed disseminated metastases to multiple organs or bones or those with any contraindications for resection received a palliative regimen of chemotherapy either capecitabine, FOLFOX (folinic acid +5 fluorouracil +oxaliplatin) or FOLFIRI (folinic acid+5 fluorouracil+irinotecan) with or without target agents.
Definitions, follow-up, statistics
In our study, metastasis is defined as the distant spread of the tumor to other organs (liver, lung, peritoneum, and others) excluding regional LNs while the LARC is under treatment with or after completion of CRT. The mean time for the detection of metastases post-CRT is the time interval between the initiation of treatment with CRT and the detection of metastases to other organs. Magnetic resonance CRM (mrCRM) is defined as the distance between the tumor tissue and the surrounding radial margin and is considered positive when it is ≤ 1 mm on MRI [12]. On the other hand, magnetic resonance tumor regression grade (mrTRG) is defined as the radiological imaging of the tumor response after CRT based on the presence of fibrosis or tumor signal intensity [13]. mrTRG1 means no evidence of tumor (complete response); mrTRG2, dense fibrosis without residual tumor (good response); mrTRG3, ≥ 50% fibrosis with the intermediate signal intensity of the tumor (moderate response); mrTRG4, obvious tumor with little fibrosis (slight response); and mrTRG5, same appearance and signal intensity of the tumor (no response) [14]. Magnetic resonance EMVI (mrEMVI) is defined as the tumor invasion of veins beyond the muscularis propria layer and is classified into 4 scores. Score 0 means no vessel close to extramural penetration of the tumor; score 1, normal caliber vessel with no tumor signal intensity; score 2, slightly expanded vessel with no signal intensity; score 3, expanded vessel with intermediate signal tumor intensity; and score 4, irregular vessel contour by tumor signal intensity. mrEMVI is considered negative for scores 0, 1, and 2 and positive for scores 3 and 4 [15].
Routine follow-up strategy was not conducted in our patients because some of them demonstrated early metastasis while others showed metastasis late in the waiting period; this would lead to changes in the follow-up schedule. Despite this, during the follow-up visits, serum CEA level, abdominopelvic CT, and chest CT were performed for the patients while total colonoscopy, positron emission tomography CT scan, and pelvic MRI were only performed in cases of recurrence.
The data for 14 patients were analyzed using IBM SPSS Statistics ver. 25 (IBM Corp., Armonk, NY, USA). The variables had been expressed as the number of patients (percentage), median (range), or mean± standard deviation.
RESULTS
Patients’ characteristics
In the study period, 1,092 patients received CRT, of whom 14 patients (1.3%) met the inclusion criteria were enrolled. The median age of the patients was 63 years (range, 41 to 80 years) with male predominance (11 [78.6%] vs. 3 [21.4%]). The median distance of the tumor from the anal verge was 7 cm (range, 5 to 10) with 8 patients (57.1%) of clinical T3 (cT3) stage and 6 (42.9%) of cT4 patients. The median pre- and post-CRT CEA levels were 4.2 ng/mL (0.8 to 95 ng/mL) and 4.7 ng/mL (0.7 to 884 ng/mL), respectively. With regard to tumor pathology, more than 57% of the tumors were moderately differentiated adenocarcinoma followed by well-differentiated adenocarcinoma (28.6%), as shown in Table 1.
Initial image (pre-CRT) of the tumors
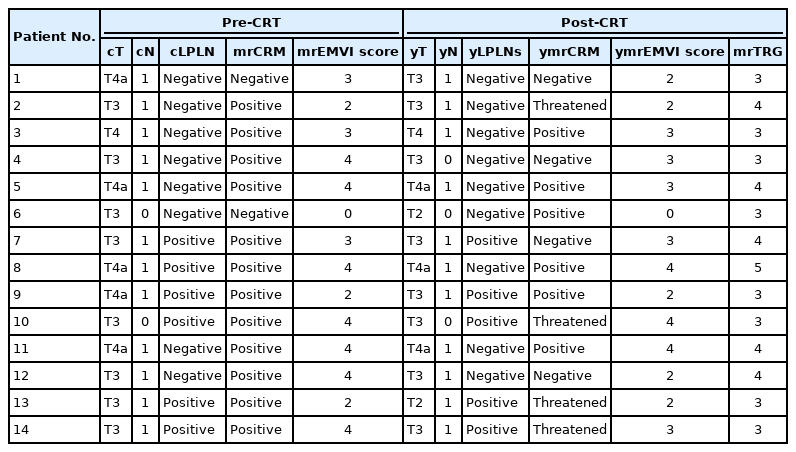
Table 2 shows the initial radiological characteristics of the tumors. mrCRM, lateral pelvic LNs (LPLNs), and mesorectal LNs were positive in 12 (85.7%), 6 (42.9%), and 12 patients (85.7%), respectively. mrEMVI was positive in 10 patients (71.4%) (score 3 in 3 cases and score 4 in 7 cases).
Pre- vs. post-CRT staging
After CRT, 3 out of 12, 2 out of 10, and 1 out of 6 patients converted from positive to negative CRM, EMVI, and LPLNs, respectively, as shown in Fig. 1. Conversely, only 4 patients showed downstaging in the T stage. Regarding the tumor response, mrTRG3 (moderate response) was detected in 8 patients while mrTRG4 (slight response) and mrTRG5 (no response) were noticed in 5 and 1 patients, respectively, as demonstrated in Table 3.

Magnetic resonance imaging for 2 patients who showed a response after chemoradiotherapy (CRT). (A) Initial (left panel; positive mrEMVI) and posttreatment (right panel; negative ymrEMVI) images of patient 1. (B) Initial (left panel; positive mrCRM and mrEMVI) and posttreatment (right panel; negative ymrCRM and ymrEMVI) images of patient 12. The arrows show the status of CRM and/or EMVI before and after treatment with CRT. mr, magnetic resonance; EMVI, extramural venous invasion; y, post-CRT; CRM, circumferential resection margin.
Metastases and methods of intervention
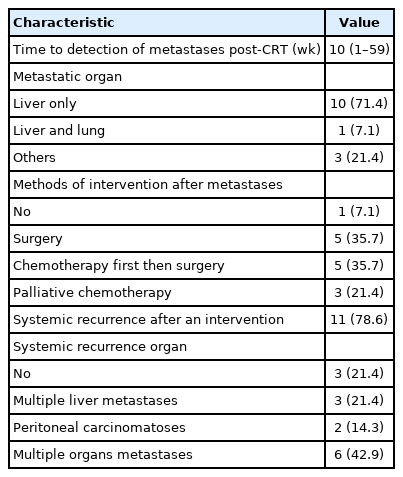
Table 4 shows that the median time for the detection of metastases post-CRT was 10 weeks (range, 1 to 59 weeks), and the liver was the commonest site of metastasis (10 cases, 71.4%). Furthermore, 1 patient developed metastases in both the liver and the lung, whereas metastases were detected in the liver with the small intestine, the liver with the peritoneum, and the liver with the lung and the peritoneum in 3 patients, respectively.
After metastases, 1 patient refused any intervention while 10 (71.4%) underwent combined surgery (5 received direct surgery and the other 5 were administered chemotherapy first followed by surgery). Regarding those who underwent combined surgery, 8 out of 10 received low anterior resection (LAR) (2 open, 5 laparoscopic, and 1 robotic) and the other 2 patients underwent open as well as laparoscopic ultra-LAR. Meanwhile, 3 patients (21.4%) received palliative chemotherapy owing to multiple liver metastases in both lobes. Eleven patients (78.6%) presented with progressive disease after intervention in the form of local and/or systemic recurrence; they comprised 3 (21.4%), 2 (14.3%), and 6 (42.9%) with recurrence in the liver, peritoneum, and multiple organs, respectively, as shown in Table 4.
DISCUSSION
Nowadays, preoperative CRT followed by TME after an 8- to 10-week gap is considered the optimal treatment modality for LARC with favorable local control; however, distant metastases occurring during or after the completion of treatment is the principal mode of failure [11].
In our case series, metastases developed in 14 out of 1,092 patients who received preoperative CRT, constituting 1.3% of the total patient population. This is a very small number that suggests that metastases after CRT may be a rare entity. This result is consistent with those reported by Choi et al. [16], who conducted a study for early metastases on 107 patients who received preoperative CRT and only 7 patients (6.5%) developed metastases during the waiting period.
In our study, patients who developed early metastases received capecitabine frequently as a radiosensitizer. This does not mean that it is a weaker chemotherapeutic agent but inversely its effect had been approved by multiple studies [17-19]. This result may be attributed to the small number of our patients; hence, further studies with a larger number of cases are recommended.
Eleven patients in our study developed systemic metastases after the intervention, of whom 6 developed multiple organ metastases and 3 presented with multiple liver metastases after they received a combined surgery of TME with wedge resection of the liver. Meanwhile, 2 patients developed peritoneal carcinomatosis after they underwent ultra-LAR with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. These results denote a poor prognosis for patients who developed early metastasis after CRT and were managed by any type of intervention.
In our series, we obtained a non-good or complete response after CRT according to the mrTRG score and this is considered a risk factor for metastasis [20]. This may be due to the high rate of positive mesorectal LNs, positive mrCRM, positive mrEMVI, and moderately differentiated adenocarcinoma, which are the chief risk factors for metastasis [21, 22].
Consequently, despite the median duration of 10 weeks after CRT, only 4 patients (28.5%) showed downstaging in the T stage which was less than the proportion of patients (41.2%) reported by Yeo et al. [23]. Contrarily, 12 (85.7%) of our patients presented with involved mrCRM; this is a higher rate than that reported in the available literature (4% to 18%) [24] and only 3 of them (25.0%) converted to free CRM after CRT. This low rate of response and a higher rate of involvement may be considered the cause of metastases [25], poor prognosis, and relapse [26].
In a previous study [27] comparing positive and negative mrEMVI, the risk of distant metastasis was 4 times increased in positive cases (52%) than that in negative ones (12%). In our study, the rate of baseline mrEMVI-positive status was high (71.4%) and the response to CRT was slight to moderate (2 cases only out of 10 with mrTRG 3 and 4). Our results support the poor prognosis of patients with initial positive mrEMVI and their poor response to CRT. This group of patients may be considered resistant to the standard CRT regimen; hence, other treatment strategies such as induction or consolidation chemotherapy should be considered for those patients.
To date, multiple questions have been asked about patients who will not respond to preoperative CRT and who will develop early metastases either during or after completion of treatment [16]. Therefore, from a practical point of view, changing the chemotherapy regimen and/or radiation treatment dose with a short and meticulous follow-up period may help in detecting non-responders early and give a chance to modify the treatment protocol.
Our study has the limitations of being a retrospective study with a small number of cases. Nevertheless, to our knowledge, our study is the first to summarize the characteristics of patients presented with newly developed metastases after completion of CRT and before surgery.
In conclusion, although metastasis after CRT is a rare entity, it should not be neglected. In our study, patients who developed metastases after CRT had a middle third LARC with grade II adenocarcinoma, and the liver was the commonest site. Patients with advanced status which were positive mesorectal LNs, positive mrRCM and positive mrEMVI should be closely evaluated in restaging period. Despite a long waiting period after CRT, poor responder can show the metastases during restaging period. Further studies with a larger number of patients are needed to treat those patients who showed early metastases after CRT.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.